Coenurosis is a cystic parasitic disease affecting ruminants and other mammals worldwide. The disease is caused by Coenurus cerebralis, the larval stage of the tapeworm Taenia multiceps. Sheep and goats are frequently infected, while cases in cattle are rare. Here we describe the first recorded case of bovine coenurosis in North Macedonia. The diagnosis was based on the neurological clinical signs and the postmortem findings of a 1-year-old bull with symptoms typical for coenurosis, i.e. ataxia, circling movements, mild depression and impaired vision. Postmortem, a cyst was found in the left cerebral hemisphere and was confirmed as fertile Coenurus cerebralis by parasitological analysis. These findings suggest that coenurosis should be considered part of the differential diagnosis in cattle with neurological symptoms. This highlights the need for routine surveillance of this disease in livestock and proactive tracking of the parasite in the final hosts to enhance disease management.
Coenurosis is a parasitic disease caused by the larval stage of the tapeworm
Taenia multiceps (Leske 1780), affecting the central nervous system of sheep, other domestic and wild ruminants, horses, pigs and humans (
1, 2). The adult tapeworm parasitises the small intestine of canids (definitive hosts) and is excreted as gravid proglottids or eggs in the faeces. The intermediate hosts (ruminants, horses, pigs) and humans (accidental hosts) become infected by ingesting eggs through contaminated food, water or pasture. Oncospheres are then released in the intestine, penetrate the mucosa, enter the bloodstream, and eventually reach the brain and spinal cord, rarely other tissues, where the larval stage (
Coenurus cerebralis) develops (
3, 4). The fully developed
Coenurus cerebralis appears as a 5-6 cm cyst filled with white fluid surrounded by a transparent membrane. The cyst is filled with 400-500 invaginated protoscoleces organised into 1-23 clusters visible as white dots connected to the membrane. The life cycle completes in 40-60 days when the definitive hosts ingest the fertile coenurus with mature protoscoleces (
5, 6).
The infected intermediate hosts develop symptoms that vary depending on the location of the cyst, its size and the level of brain compression. The clinical manifestations are characterised by moderate depression, incoordination, seizures, circling, and total or partial loss of vision (
7, 8). The economic losses are associated with decreased or ceased production, compromised genetic value and death of the infected animals (
1).
Coenurosis has been extensively reported in sheep and goats worldwide. The disease is prevalent mainly in rural regions, where dogs and sheep are in close contact and share the same environments. In cattle, the disease is uncommon (
2, 9), and despite its potential impact on animal health and production, there have been a few reports in certain regions (
1).
Coenurosis is prevalent among sheep in North Macedonia and has also been detected in goats (personal communication with local veterinarians). However, there have been no reported cases of the disease in cattle. The present study aims to report and describe the first case of bovine coenurosis in North Macedonia.
CASE DESCRIPTIONThe affected animal was a 1-year-old Simmental bull with a history of neurological symptoms, bred extensively on a farm in a village in the central part of North Macedonia. The farm has 50 cattle and 1,000 sheep bred in a free-range system. The animals spend most of their time together, sharing the same pasture during the year. Ten shepherd dogs also live freely on the same farm. According to the farm owner, the dogs have never been dewormed and are often fed with the remains of slaughtered livestock (from both sheep and cattle), including the brain. The sheep on the farm have a long history of coenurosis, and only a few have had their brain cysts surgically removed.
The farm owner noticed that the 1-year-old bull had decreased growth in the last month (March 2023) and started to manifest clinical signs similar to those in sheep with coenurosis. He alerted the family veterinarian, who examined the bull.
The animal had a normal appetite and rectal temperature but was slightly depressed and ataxic. The most characteristic clinical signs were circling movements (to the left) and frequent hitting of obstacles. The veterinarian suspected coenurosis and euthanised the bull due to poor prognosis.
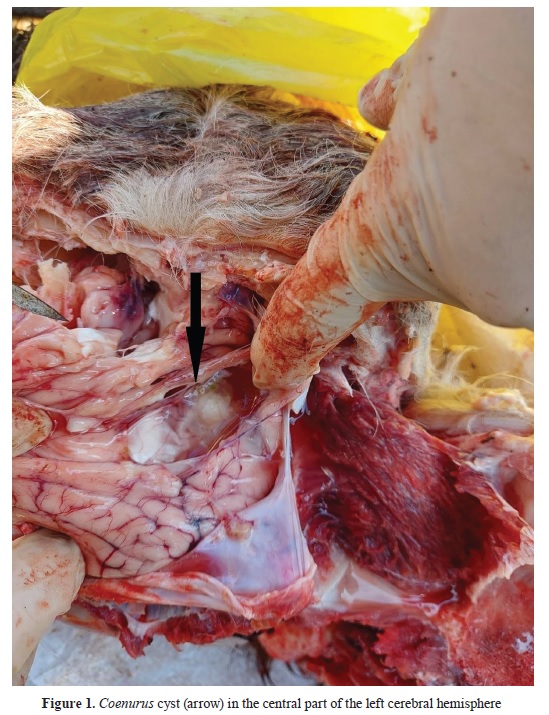
At necropsy, a cyst containing clear fluid and protoscoleces was detected in the central part of the left cerebral hemisphere, under the meninges and a layer of brain tissue (
Fig. 1). The bone above the cyst was softened, and the skin was bulged.
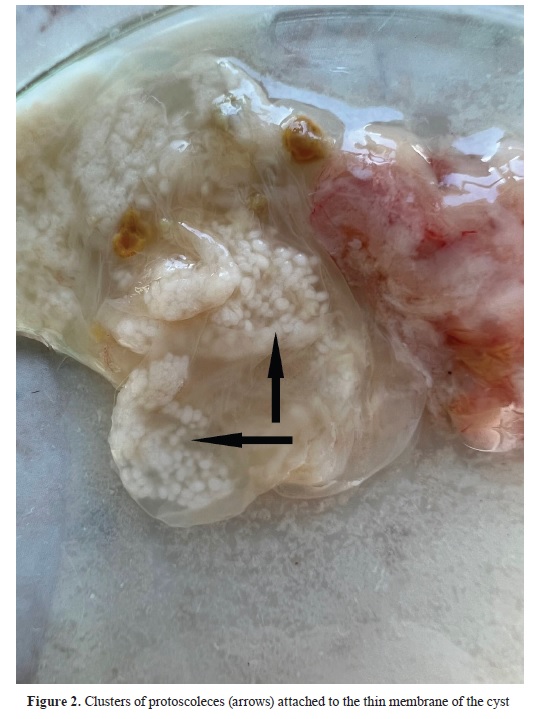
The parasitological examination revealed that the cyst was large (5 cm in diameter), translucent and with numerous clusters of protoscoleces attached to the internal membrane (
Fig. 2). The morphology of the cyst was typical for
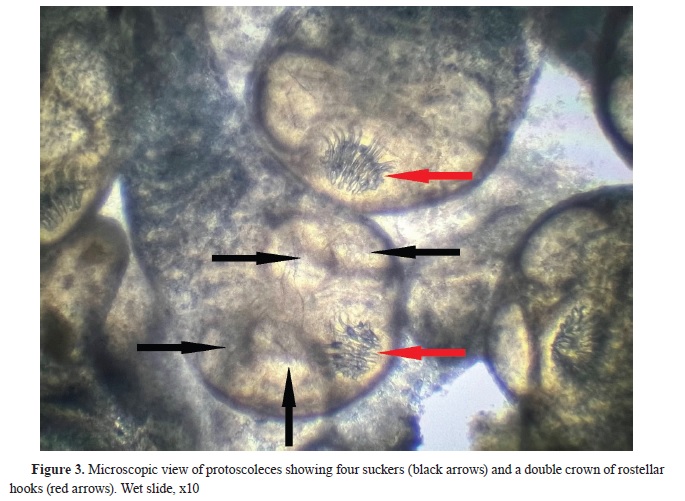
Coenurus cerebralis. The microscopic analysis of the protoscoleces revealed the presence of four suckers and a double crown of 28 rostellar hooks in each scolex (
Fig. 3). The large hooks ranged from 140 to 150 μm, while the small hooks measured between 90 and 100 μm in length. The protoscoleces were actively moving and the cyst was declared fertile.
 DISCUSSION
DISCUSSIONThis study reports the first finding of
Coenurus cerebralis in cattle in North Macedonia. The bull had neurological symptoms consistent with coenurosis and was euthanised due to poor prognosis. The disease was confirmed with postmortem and parasitological analyses. The morphological characteristics of the cyst found in the left cerebral hemisphere resembled
Coenurus cerebralis.
Coenurosis is a cosmopolitan disease in small ruminants, but reports of bovine coenurosis are rare in the scientific literature. In case of a cattle, the disease has been reported in various countries, including Great Britain (
10), Japan (
11), Iran (
12), Bangladesh (
13), Bulgaria (
14), Greece (
2, 9), Turkey (
3, 7, 8), Uruguay (
15), Brazil (
16, 17) and Italy (
1, 4, 5). There have been even fewer reports on the prevalence of bovine coenurosis. The incidence of coenurosis in calves in Bangladesh was 2.47% (13), and the prevalence of bovine coenurosis in the Eastern Anatolian region of Turkey was only 0.47% (
3). Despite the sporadic nature of these reports, the presence of bovine coenurosis in different regions of the world raises concerns about its potential impact on animal health and productivity. As this is the first report, the prevalence of the infection within the bovine population in North Macedonia remains to be determined. The cyst matures in 7-8 months (
1), which implies that the prevalence studies should focus on cattle around the age of one year.
The location and size of the cyst have an essential role in the pathogenesis of the disease (
3, 7). The brain and the spinal cord are predilection sites for the cyst location. The cysts have been reported in different parts of the cerebrum (
2, 11, 12, 13), spinal cord (
13) and cerebellum (
7, 9, 13, 14). In our case, the cyst was located in the cerebrum, the most frequent occurrence in bovine coenurosis cases. The cyst was 5 cm in diameter, conforming to the typical size of a coenurus cyst reported previously (
1, 14).
Bovine coenurosis manifests with incoordination and impaired vision (
12), including uncontrolled movements, ataxia, limb paralysis, circling, exhaustion, and, ultimately, death (
14). The bull from our case had ataxia, mild depression, circling movements (to the left) and was frequently hitting obstacles, which is a sign of impaired vision.
The diagnosis of coenurosis is challenging and can be mistaken for other similar diseases and conditions like listeriosis, bovine spongiform encephalopathy, rabies, vitamin B1 deficiency, brain abscess and cerebral echinococcosis. For the correct diagnosis, the veterinarian must consider the age of the animal, clinical signs, and the results of diagnostic imaging and post-mortem examinations (
1, 6). In our case, the diagnosis was based on the clinical signs typical for coenurosis and the postmortem finding of the cyst in the cerebrum. The parasitological analysis aided the diagnosis and confirmed the cyst as
Coenurus cerebralis. The cyst was fertile, suggesting that it was capable of causing an infection if fed to the dogs on the farm. The farmer had a similar case with a bull the previous year, but he did not report it and slaughtered the animal. The head was given to the dogs.
The control of coenurosis poses significant challenges and has thus far yielded unsatisfactory results. Once the disease has been diagnosed, the prognosis is unfavourable, as the typical outcome is death. Preventive strategies are the most efficacious means of controlling coenurosis. The control can be accomplished by implementing a consistent deworming of farm dogs with praziquantel (5 mg/kg body weight) administered every 6-8 weeks. The treatment is effective for both
Taenia and
Echinococcus spp., as dogs, especially farm dogs, can be co-infected with different tapeworms. Additionally, it is crucial to appropriately dispose of sheep and cattle carcasses to prevent them from being scavenged by dogs and wild carnivores (
1). In our case, the farm owner had never dewormed the dogs and regularly fed them with sheep and cattle carcasses. He was informed that in this way, he is contributing to the spread of the disease and that he has to implement control measures to minimise the losses. Additionally, the owner was warned that coenurosis poses a substantial zoonotic threat as it has the capacity to induce severe and potentially fatal complications in human beings (
18). Humans, particularly children, can serve as accidental hosts for the parasite when they unintentionally contract the infection through the ingestion of eggs from the faeces of definitive hosts (
19) or via the foodborne pathway by ingesting eggs from contaminated fruits and vegetables (
20).
CONCLUSIONOur results indicate that bovine coenurosis is present in North Macedonia. Although rare, the infection can be a serious cause of neurological disease in cattle. This report highlights the importance of considering coenurosis in differential diagnosis of cattle with neurological symptoms. We recommend that veterinarians be aware of the possibility of bovine coenurosis and be familiar with the clinical signs and diagnostic approach for the disease.
As there are no data for the presence of
Taenia and
Echinococcus spp. in the dog population in our country, this report also suggests the need to initiate a new study on the surveillance of
Taenia multiceps (including other
Taenia and
Echinococcus spp.) in carnivores, with particular emphasis on farm dogs.
CONFLICT OF INTERESTThe authors declare that they have no potential conflict of interest with respect to the authorship and/or publication of this article.
ACKNOWLEDGMENTSThis research was supported by the Faculty of Veterinary Medicine–Skopje.
AUTHORS’ CONTRIBUTIONSAC conceived the study, supervised the parasitological testing, contributed to interpreting the results and wrote the manuscript. LR, AT, IS and IC performed the parasitological testing and interpreted the results. BD and PD performed the clinical and postmortem examination and interpreted the results. JS supervised the parasitological testing and contributed to the interpretation of all results. All authors have revised and approved the final version of the manuscript.

 10.2478/macvetrev-2023-0029
10.2478/macvetrev-2023-0029

