Intestinal parasites in shelter dogs present significant health risk to animals and humans. This cross-sectional study examined the prevalence, risk factors, and zoonotic implications of intestinal parasites in 156 shelter dogs across all eight regions of North Macedonia. Fecal samples were analyzed using fecal smears and flotation techniques. The overall prevalence was 70.51%, with hookworms (Ancylostomatidae) (36.54%), Giardia spp. (24.36%), and Trichuris spp. (21.79%) identified as the most prevalent parasites. Significant risk factors included young age, mixed breed, and regional variations. Although the treatment reduced infection rates, regional disparities remained. These findings underscore the urgent need for standardized deworming protocols and One Health strategies to mitigate zoonotic risks
INTRODUCTION
Intestinal parasites represent a significant global health concern for dogs, with prevalence notably higher in shelter populations compared to owned dogs (
1,
2). Shelter environments often provide conditions conducive to parasite transmission, including high population density, varying healthcare standards, limited resources, and increased host stress, which can elevate susceptibility (
3). Consequently, infection rates in shelter dogs worldwide can exceed 70% (
4), contrasting with rates as low as 3.5% for intestinal helminths in household pets (
2).
Many canine parasites, such as
Giardia spp.,
Toxocara canis, and Ancylostomatidae (
Ancylostoma spp. and
Uncinaria stenocephala), possess zoonotic potential, thereby increasing the epidemiological significance of infections originating in shelters (
3). These specific parasites often show dramatically higher prevalence in shelters (e.g.,
Giardia spp. >40% vs. 15% in owned dogs;
T. canis ~65% vs. 15%, respectively) (
5,
6). High rates are well- documented in the Balkan region, supported with studies from Serbia, Bulgaria, Albania, and Greece which consistently report higher parasite loads in shelter dogs, often linked to overcrowding and hygiene challenges (
6,
7,
8,
9). The zoonotic risk is particularly relevant due to the close contact between shelter animals and humans, posing a significant risk, especially to children and immunocompromised individuals (
3,
10).
Several host-related risk factors influence the prevalence of intestinal parasitism in dogs. Age is a primary determinant, with younger dogs often showing increased vulnerability due to their immature immune systems and specific transmission routes (
3,
11). The influence of a dog’s sex is considered a potential risk (
12,
13), though findings on its direct association with infection rates are conflicting (
9). Likewise, the role of breed has been investigated, but results regarding its significance as a risk factor remain inconsistent in the literature (
5,
14,
15).
Climate significantly dictates canine parasite prevalence by influencing parasite life cycles, often resulting in higher transmission rates in favorable tropical conditions and seasonal variations (
10,
16). Geographic location further modulates risk, with urban dogs typically showing lower infection rates due to better veterinary access compared to rural dogs (
17), although urban parks can still represent contaminated environments (
18).
Treatment protocols for parasitic infections in shelter environments have shown variable efficacy. Factors, including parasite species, host characteristics, and environmental conditions, may influence the effectiveness of these interventions (
3). However, systematic evaluations of treatment outcomes across different regional and management settings remain scarce. The growing global concern of anthelmintic resistance, especially in high- transmission settings such as animal shelters, poses challenges to parasite control and highlights the importance of continuous monitoring to maintain the efficacy of treatment strategies (
3).
Based on the previous researches and the unique challenges of shelter environments, we hypothesized that the prevalence and species distribution of intestinal parasites in shelter dogs in North Macedonia are significantly influenced by regional, demographic, and environmental factors. The current treatment protocols, as well, vary in effectiveness, thereby necessitating standardized control measures to mitigate zoonotic risk.
MATERIAL AND METHODS
Study design and data collection
This cross-sectional study is based on collected data from 156 shelter dogs across all eight regions of North Macedonia between September 2023 and October 2024. Samples were collected as follows: 7.05% (11/156) from the Vardar region, 10.89% (17/156) from the Southwestern region, 30.12% (47/156) from the Southeastern region, 2.56% (4/156) from the Eastern region, 1.92% (3/156) from the Northeastern region, 9.61% (15/156) from the Pelagonia region, 9.97% (14/156) from the Polog region, and 28.84% (45/156) from the Skopje region. The dogs were housed in individual cages with concrete walls and floors, which were regularly cleaned and maintained. The study population consisted of newly admitted stray dogs and dogs that had resided at the facilities for several years. Standard intake procedures for all dogs involved a clinical examination, deworming, rabies vaccination, and neutering/spaying. Following these steps, the animals were either placed for adoption or returned to their original locations. Their diet was composed of commercial dog food.
Fecal samples were collected from the surface layer of the feces in the cages using pre-labeled bags to prevent contamination. The samples were placed in styrofoam containers with cooling packs and transported to the Laboratory for Parasitology and Parasitic Diseases at the Faculty of Veterinary Medicine in Skopje on the same day. All fecal samples were analyzed within 24 h of collection.
For each dog, we recorded demographic information (age, sex, and breed), location data (region, shelter), parasitological examination results, and treatment history. Age was categorized into three groups: young (≤2 years), middle-aged (3-5 years), and older (>6 years) dogs. Breed status was classified as either mixed or purebred based on phenotypic characteristics.
Parasitological examination
All fecal samples were macroscopically examined for consistency, odor, color, and evidence of blood, mucus, pus, undigested food, or parasitic elements. Two complementary techniques were employed for microscopic analysis: direct fecal smear and flotation.
For the direct fecal smear, a small sample (approximately 0.5 g) was placed on a microscope slide with several drops of physiological saline, thoroughly homogenized, and covered with a coverslip for immediate examination. The flotation technique involved combining approximately 3 g of fecal material with 50 mL of zinc sulfate solution (specific gravity: 1.2), which was the only saturated solution used in this study. This mixture was homogenized and filtered through a 250 μm metal sieve mesh into 15 mL centrifuge tubes. The tubes were filled with zinc sulfate solution until a convex meniscus formed at the rim, then covered with a coverslip and centrifuged at 1500 RPM for 10 min. The coverslip containing concentrated parasitic elements was carefully transferred to a microscope slide following centrifugation.
All prepared slides were systematically examined under a light microscope at multiple magnifications (40x, 100x, and 400x). Parasites were identified to genus or species level according to established morphological criteria (
19).
Statistical analysis
The data were prepared using Microsoft Excel and analyzed using R statistical software (version 4.4.3; R Foundation for Statistical Computing, Vienna, Austria). The significance level was set at 0.05 for all statistical tests, and 95% confidence intervals were calculated where applicable.
The initial descriptive statistics were calculated for all variables. Categorical data were presented as frequencies and percentages, while continuous data were expressed as means ± standard deviations.
Regional differences in infection rates were assessed using chi-square tests of independence. When expected cell frequencies were less than 5, Fisher’s exact test was employed. Effect sizes for chi-square/Fisher’s tests were quantified using Cramér’s V and post-hoc power was calculated for the main comparisons.
A multivariate logistic regression model was constructed to identify significant risk factors for parasitic infection. The model included age, sex, breed status, and region as predictors. Interaction terms were tested and included if significant (p<0.05). The strength of associations (effect sizes) was quantified using odds ratios (OR) with 95% confidence intervals. The model fit was assessed using the Hosmer-Lemeshow goodness-of-fit test and the area under the receiver operating characteristic curve (AUC-ROC).
The treatment effectiveness was evaluated by calculating risk differences and risk ratios between treated and untreated groups. The Number Needed to Treat (NNT) was calculated as the reciprocal of the absolute risk reduction, with 95% confidence intervals. Risk differences, risk ratios (RR), and the number needed to treat (NNT) with 95% CI were calculated as measures of effect size, with Cohen’s
h reported for the overall treated vs untreated comparison. Stratified analyses were performed to assess treatment effectiveness by region and parasite type.
Associations among different parasite species were evaluated using odds ratios and chi-square tests. The Bonferroni correction was applied for multiple comparisons to maintain the family-wise error rate at 0.05.
Post-hoc power calculations (α=0.05) were performed for the main analyses, including regional differences in infection rates, infection type distribution, and treatment effectiveness, to evaluate the sensitivity of the study to detect true effects.
Use of AI-assisted technologies
The authors used Grammarly platform (
https://grammarly.com) solely for the purpose of enhancing the linguistic quality of the manuscript, specifically focusing on grammar, punctuation, sentence structure, and overall writing style. The platform was not used in any aspect of the scientific content, including the development of the methodology, analysis of results, or formulation of conclusions, which remain entirely the original work of the authors. The authors accept full responsibility for the accuracy and integrity of the manuscript, including any information processed or refined using AI-assisted technologies.
RESULTS
Identified parasite species
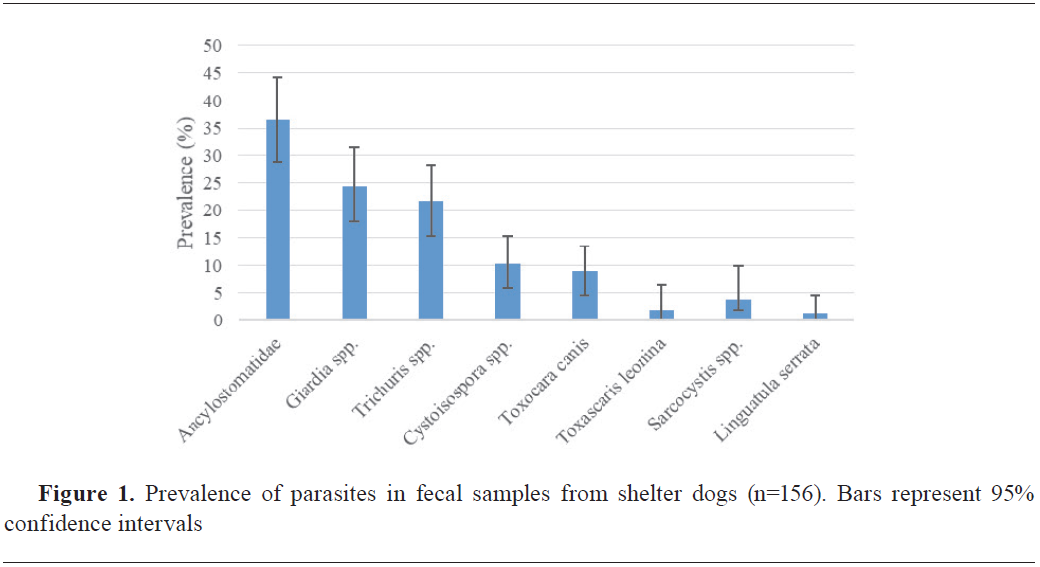
Of the 156 fecal samples analyzed, a significant overall prevalence of parasitic elements was detected at 70.51% (110/156, 95% CI: 63.36%-77.67%). The most common parasites were hookworms (36.54%), followed by
Giardia spp. (24.36%), and
Trichuris spp. (21.79%),
Cystoisospora spp. (10.26%),
T. canis (8.97%),
Toxascaris leonina (1.92%), S
arcocystis spp. (3.85%), and
Linguatula serrata (1.28%). No cestode eggs were detected in the tested samples (
Fig. 1).
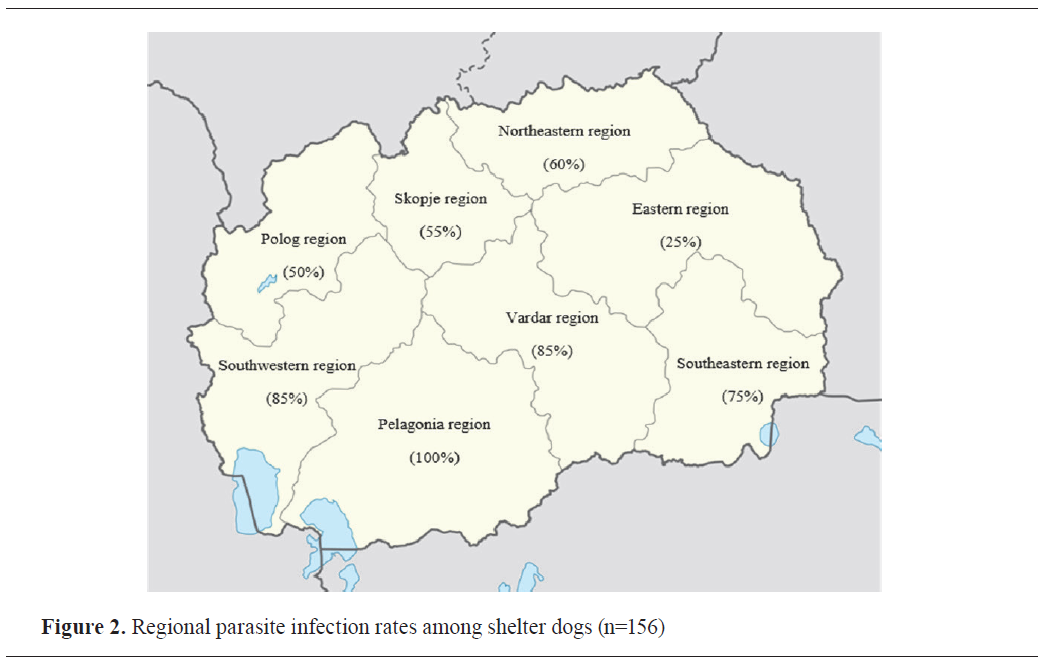
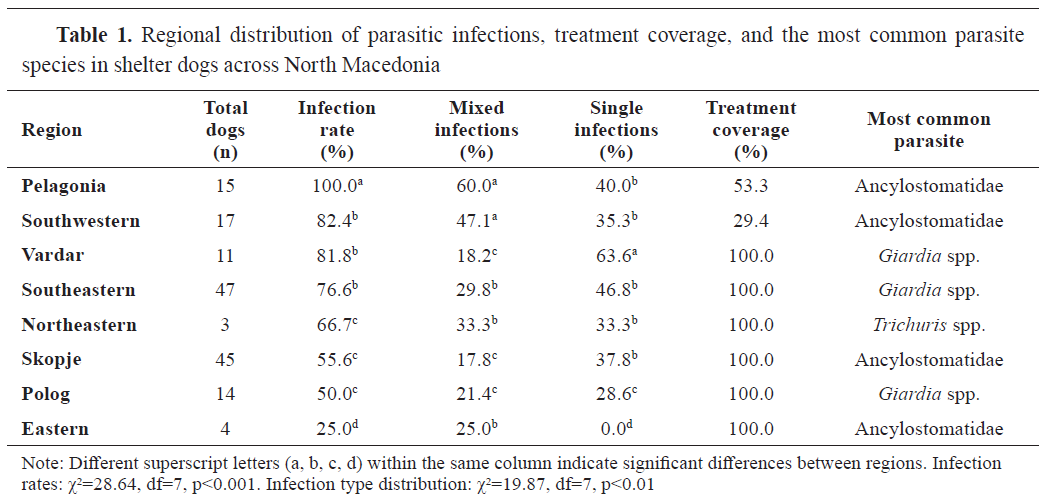
Regional distribution of parasitic infections
Significant regional variations were observed in parasite infection rates (χ²=28.64, df=7, p<0.001, Cramér’s V=0.428,
post hoc power = 0.99) (
Table 1). The highest infection rates were recorded in the Pelagonia region (100%), where all fecal samples tested positive for at least one parasite. In contrast, the Eastern region showed the lowest infection rate (25%) (
Fig. 2).
 Infection type distribution
Infection type distribution
Single-parasite infection was detected in 41.02% (64/156, 95% CI: 33.30%-48.74%) of the samples. Mixed infection was detected in 29.49% of the samples (46/156, 95% CI: 22.33%-36.64%), specifically involving two (21.15%, 33/156, 95% CI: 15.20%-28.18%), three (6.41%, 10/156, 95% CI: 3.47%-11.59%), and four (1.92%, 3/156, 95% CI: 0.66%-5.48%) species of parasites.
Coinfection analysis revealed significant associations between Ancylostomatidae and
Trichuris spp. (8.95%, 14/156, OR=2.84, 95% CI: 1.46%-5.52%, p<0.01). Odds ratio (OR=2.84, 95% CI: 1.46–5.52) served as the effect size for the coinfection association. Lower prevalence co-infections included: Ancylostomatidae with
Giardia spp. (2.56%, 4/156); Ancylostomatidae with
T. canis (1.92%, 3/156); Ancylostomatidae with
Cystoisospora spp. (1.92%, 3/156); and a triple co-infection of Ancylostomatidae,
Giardia spp., and
Trichuris spp. (1.28%, 2/156). Other co-infections occurred at a prevalence below 1%.
Mixed infections were particularly prevalent in the Pelagonia region (9/15, 60% CI: 35.75%-80.18%), while single-parasite infections dominated in the Vardar region (7/11, 63.64%, 95% CI: 35.38%-84.84%). The distribution of single versus mixed infections showedsignificantregionalvariation(χ²=19.87, df=7, p<0.01, Cramér’s V=0.357,
post-hoc power=0.9).
The distribution of infection types showed significant age-dependent patterns, with young dogs showing the highest proportion of single infections (38/78, 48.72%, 95% CI: 37.95%-59.61%), while middle-aged and older dogs showed more balanced distributions between single and mixed infections. The association among age group and infection type showed a negligible effect size (Cramér’s V=0.012) and very low
post-hoc power (0.05), indicating limited sensitivity to detect differences across age groups. The regional variations in infection type distribution were significant (p<0.01), with distinct patterns observed across different geographical areas (
Table 1).
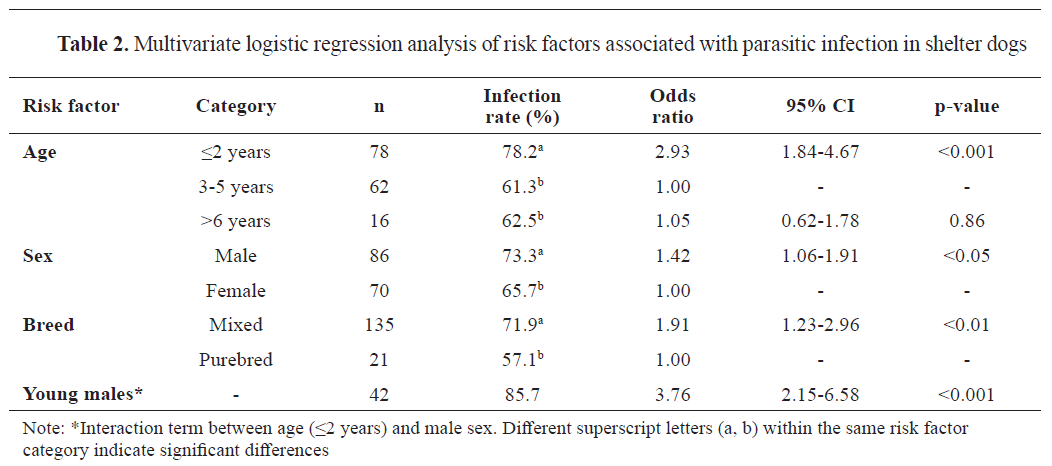
Demographic risk factors and multivariate analysis
The multivariate logistic regression identified several significant risk factors for parasitic infection (
Table 2). Age emerged as the strongest predictor, with young dogs (≤2 years) showing significantly higher infection rates compared to middle-aged dogs (OR=2.93, 95% CI: 1.84%-4.67%, p<0.001).
The association among age group and infection type showed a negligible effect size (Cramér’s V=0.012) and very low
post-hoc power (0.05), indicating limited sensitivity to detect differences across age groups. A significant interaction was observed between age and sex, with young males showing the highest risk (OR=3.76, 95% CI: 2.15%-6.58%, p<0.001). Mixed-breed dogs, comprising 87% of the study population, showed higher susceptibility to infection than purebred dogs (OR=1.91, 95% CI: 1.23%-2.96%, p<0.01). This effect was most pronounced in young dogs, where the protective effect of purebred status was strongest. Odds ratios served as effect sizes in the multivariable models; model performance was supported by AUC-ROC=0.75 and the Hosmer–Lemeshow test (p>0.05), indicating acceptable discrimination and model fit.
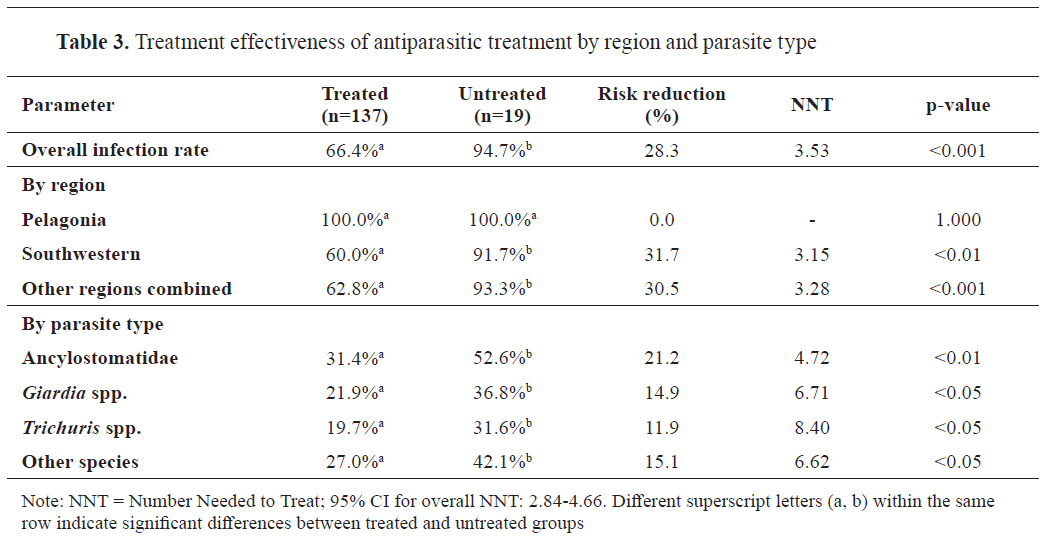 Treatment effectiveness
Treatment effectiveness
The treatment effectiveness analysis demonstrated a significant reduction in infection rates among treated dogs compared to untreated dogs (66.4% vs 94.7%, p<0.001, Cohen’s
h=0.772,
post hoc power=0.88), with an overall risk reduction of 28.32% (95% CI: 21.45-35.19). The number needed to treat (NNT) varied by parasite species and region, with the lowest NNT observed in the Southwestern region (3.15) for all parasites combined. Treatment effectiveness showed significant variation by parasite species, with the highest efficacy against Ancylostomatidae (risk reduction=21.2%, p<0.01). Notably, treatment effectiveness could not be evaluated in the Pelagonia region, where the prevalence was 100% (
Table 3). For treatment outcomes by region and parasite type, effect sizes were expressed as risk reduction and number needed to treat (NNT), which provide clinically interpretable measures of effect.
DISCUSSION
This study assessed the prevalence of intestinal parasites in fecal samples from dogs housed in public shelters in the Republic of North Macedonia. The overall parasite detection rate of 70.51% observed in this study aligns with findings from most global studies, including those conducted in the Balkan region (
6,
7,
8,
9).
In this study, hookworms (Ancylostomatidae) were the most frequently detected parasites (36.54%), identified alone or in coinfections. Their cosmopolitan distribution is facilitated by multiple transmission pathways and they pose a recognized public health threat causing conditions like cutaneous larva migrans, eosinophilic enteritis, or neuroretinitis in humans (
20). The high prevalence observed strongly suggests significant environmental contamination and efficient transmission within the shelter environment where close contact and shared spaces promote parasite transmission (
3,
14,
20).
Comparatively,
T. canis was detected at a lower rate (8.97%). While both
T. canis and Ancylostomatidae are widely regarded as the most common canine helminths globally (
21), their relative prevalence differs significantly based on geographical factors (
15,
18,
22). Nevertheless, the zoonotic implications of
T. canis are serious, including visceral and ocular larva migrans (
3). A critical factor is the exceptional resilience of
T. canis eggs in the environment (
22); thus, even an 8.97% prevalence contributes significantly to environmental contamination and poses an elevated risk to exposed populations like shelter workers (
3). Consequently, these findings strongly advocate for integrated control measures encompassing strict hygiene protocols, consistent deworming schedules in shelters, and targeted public health education (
3,
22).
Giardia spp. was one of the most frequently detected parasites. Numerous studies have examined the prevalence of
Giardia spp. in different dog populations worldwide, with results showing variability depending on factors such as age, environmental conditions, geographical region, and the animal’s health status (
3,
23). Infections with
Giardia spp. are more common in crowded facilities with poor hygiene and compromised health conditions than in dogs kept as household pets (
3).
Giardia duodenalis compromises the health of dogs and poses a significant risk to human health as it is a leading cause of diarrhea with an estimated 280 million infections annually (
24). The potential for transmission between species is a key concern; while dogs are typically infected with host-specific genotypes C and D, they can also carry human- specific genotypes, and humans in turn can be infected with genotype C (
25). This zoonotic perspective is particularly important in Southeastern Europe, where infections with
G. duodenalis in dogs are more prevalent than in Western Europe, and the presence of genotypes C and D has been confirmed in North Macedonia (
23).
The high prevalence of
Trichuris spp. aligns with global studies, which consistently report
Trichuris spp. as one of the most common intestinal parasites in shelter dogs (
1,
26). The resilience of
T. vulpis eggs in the environment contributes to the higher infection rates observed in shelter dogs. These eggs can survive in contaminated soil for extended periods, making it easy for dogs to become infected through contact with contaminated surfaces (
14). Additionally, the environmental contamination with
T. vulpis eggs poses a risk not only to the dogs themselves but also to shelter staff and potential adopters, as these parasites have been suggested to have zoonotic potential (
27).
The predominance of Ancylostomatidae and
Giardia spp. is well-known (
3,
22). However, the significant association between Ancylostomatidae and
Trichuris spp. represents an important finding that may influence treatment strategies. This coinfection pattern suggests shared environmental risk factors or potential synergistic relationships between these parasites, a phenomenon that has been observed in other studies (
1,
4). From a clinical perspective, this association requires adaptation of treatment protocols. The anthelminthic resistance profiles of these parasites often differ, with
Ancylostoma spp. showing increasing resistance to benzimidazoles, while
Trichuris spp. exhibits reduced susceptibility to macrocyclic lactones (
3,
28). Therefore, identifying this co-infection pattern should trigger the use of combination therapies with multiple mechanisms of action, such as emodepside-praziquantel or moxidectin- imidacloprid formulations, rather than single-class treatments (
27).
Cystoisospora spp. was detected in 10.89% of the samples, while
Toxascaris leonina was found in 1.92%. These parasites have a cosmopolitan distribution and show varying prevalence rates in shelter populations (
3), so their presence in North Macedonia is not unexpected.
Sarcocystis spp. was detected in a single shelter in the Southeastern region. The presence of multiple livestock slaughterhouses in this region suggests that feeding dogs’ slaughterhouse waste may be a potential source of infection. These findings are consistent with regional data, which indicate a low prevalence of
Sarcocystis spp. infections in dogs (
7,
29).
Linguatula serrata was detected in two samples only in one shelter located in the Skopje region. Data on its prevalence in Europe is limited, but cases have been reported in dogs from Romania (
30,
31) and Greece (
32). Infections with this parasite are endemic in Iran, posing a significant health risk to both dogs and humans (
33).
Cestode eggs were not detected in this study. Similar negative results for taeniid eggs have been reported in other studies (
5). Centrifugal flotation techniques are more specific for nematodes than for cestodes, which can lead to misdiagnosis of cestode infections (
3). Tapeworm eggs, including those of
Dipylidium caninum and
Taenia spp., are often difficult to detect in fecal flotation tests (
11,
34), suggesting that the prevalence reported in this study may be underestimated. Diagnosing
Echinococcus spp. is particularly challenging, with the gold standard method involving intestinal scraping and sieving of gastrointestinal contents postmortem (
34,
35). Cystic echinococcosis, primarily caused by the larval stage of
Echinococcus granulosus, poses a significant public health concern in North Macedonia. These infections are endemic in the country, with high prevalence rates in livestock, particularly in slaughtered cattle and sheep, reaching up to 60% in certain regions (
36).
We observed a high prevalence of hookworms in this study. The geographical occurrence of these parasites varies significantly by region (
2).
Uncinaria stenocephala is generally more common in cooler temperate climates, whereas
Ancylostoma caninum development is favored by warm, humid environments (
3,
15,
17). Despite its preference for warmth,
A. caninum can also be frequently found in colder areas, suggesting potential adaptation or other variables at play (
2). The morphological similarity of
Ancylostoma spp. and
Uncinaria spp. eggs necessitates supplementary diagnostic testing for precise species differentiation (
7,
20,
28).
Significant geographic variations were observed within our study area, with southern regions exhibiting higher infection rates, likely due to favorable climatic conditions. Notably, prevalence in the Pelagonia (100%) and Southwestern (82.35%) regions exceeded levels reported in comparable Balkan studies (
6,
7,
8,
9), highlighting the need for location-specific controls, although small sample sizes in Eastern/Northeastern regions require further confirmation.
Young dogs (≤2 years) showed a high infection rate (78.21%), supporting previous studies that suggest their increased vulnerability to parasitic infections. This may be due to their immature immune systems or behavioral factors (
6,
14,
16). Puppies are particularly at risk for
T. canis and
А. caninum due to transplacental and transmammary transmission (
13). Moreover, the exploratory tendencies of young dogs, such as licking contaminated surfaces and engaging in frequent social interactions, further enhance their likelihood of encountering infectious parasite stages, especially in shelter settings (
3,
14,
21). These findings require shelters to use age-specific parasite control strategies, such as frequent deworming for young dogs, treating pregnant females, separating housing areas to reduce cross-infection, and prioritizing environmental hygiene (
3).
Interestingly, young male dogs showed the highest infection rate (85.71%). While some research suggests males are more susceptible (
12), other studies indicate higher infection rates in females (
13,
37) or no significant gender differences (
5,
15). These conflicting results suggest that environmental factors and behavior may play a key role (
12,
13). Additionally, mixed-breed dogs had a higher infection rate (71.85%) than purebreds (57.14%). While some studies support this finding (
14), others suggest breed does not significantly influence parasitic infection risk (
5,
15).
The significant reduction in infection rates among treated dogs (28.32% risk reduction) demonstrates the effectiveness of antiparasitic treatment programs. However, the variation in treatment coverage across regions highlights implementation challenges that need to be addressed.
Several combinations of antiparasitic agents are used in shelters in North Macedonia, with the most common being pyrantel pamoate, and praziquantel, with or without ivermectin. Another combination includes febantel, pyrantel embonate, and praziquantel, with or without ivermectin. This broad-spectrum approach targets a variety of parasites, including nematodes, cestodes, and protozoa, ensuring comprehensive parasite elimination. While these treatments appear effective, persistent infections in some dogs suggest the possibility of reinfection or resistance, emphasizing the need for continuous deworming protocols and better hygiene practices (
3).
Resistance to multiple antiparasitic agents in
А. caninum has been confirmed in recent years (
28,
38). The high prevalence of hookworm infections among shelter dogs in North Macedonia may indicate resistance to commonly used antiparasitic combinations, posing a serious health risk to both dogs and humans. This issue has been reported in various studies, indicating a pressing need for effective management strategies. The phenomenon of resistance is often attributed to the over-use of specific drug classes, particularly benzimidazoles and macrocyclic lactones, which have been extensively utilized in canine populations (
3,
28). Anthelmintic resistance is complex and multifactorial, with genetic mutations, particularly in the β-tubulin gene, playing a key role in reduced benzimidazole efficacy (
39). The density-dependent egg production in surviving
А. caninum post-treatment may enhance resistance, as reduced competition increases egg output, accelerating resistant population growth and complicating control efforts (
40). Inadequate deworming protocols, as well, could contribute to the emergence of resistant strains, as insufficient dosing or inappropriate drug selection may fail to eliminate the parasites effectively (
3).
The high prevalence of zoonotic parasites, particularly
Giardia spp. and hookworms, presents significant public health risks. These findings highlight the importance of implementing One Health strategies for parasite control in shelters, as suggested by recent research (
3,
21). Understanding regional infection patterns can help optimize public health interventions and resource distribution.
Several limitations should be considered when interpreting our findings. The cross-sectional design provides valuable prevalence data but limits our ability to assess seasonal variations or long- term patterns in parasite distribution. The uneven distribution of samples across regions, with smaller representation from the Northeastern and Eastern regions, may have limited our ability to detect regional-specific patterns in these areas.
Additionally, our reliance on conventional microscopy may have underestimated the true prevalence of certain parasites, particularly protozoans. This is relevant for
Giardia spp., where coproantigen immunoassays often provide higher sensitivity than flotation alone (
23). Likewise, the absence of
Cryptosporidium spp. in this study is likely a reflection of these diagnostic limitations. The zinc sulfate flotation method has low sensitivity for the parasite’s small oocysts, making conventional microscopy an unreliable detection tool for this pathogen (
3). For an accurate diagnosis of these protozoa, more sensitive techniques such as immunofluorescent assays, ELISA, or PCR are required (
3). While a true low prevalence of
Cryptosporidium cannot be entirely ruled out, this finding highlights that the prevalence of certain parasites may be underestimated, and future studies should employ these advanced diagnostic methods.
The absence of molecular characterization of parasites, particularly for
Giardia spp. and Ancylostomatidae, prevented us from determining the zoonotic potential of specific isolates and assessing genetic markers of resistance. Future studies would benefit from incorporating molecular techniques to identify species, assemblages, and resistance genes, providing more targeted public health recommendations.
Despite these limitations, our study provides the first comprehensive assessment of intestinal parasites in shelter dogs across North Macedonia, establishing an essential baseline for future monitoring and intervention efforts. Future research should focus on long-term monitoring, molecular diagnostics, and environmental risk assessments to develop more effective parasite control strategies. Addressing these gaps will be key to reducing the health risks of parasitic infections in shelter populations and the broader community.
CONCLUSION
This study revealed a high prevalence of intestinal parasites in shelter dogs in North Macedonia, underlining the urgent need for improved control measures. With over 70% of dogs testing positive for at least one parasite, dogs and humans have a high health risk, given the zoonotic nature of several detected species, including Ancylostomatidae and
Giardia spp.
Regional, demographic, and environmental factors significantly influence infection rates, with younger dogs, mixed breeds, and specific geographic areas exhibiting higher susceptibility. The study also detected treatment effectiveness but noted regional disparities, underlining the need for standardized deworming protocols and improved hygiene practices within shelters.
Given the potential for parasite transmission to humans, especially in shelter environments with close human-dog interactions, implementing a One Health approach is crucial. Following the internationally established guidelines and incorporating routine screening, targeted treatments and better sanitation measures will contribute to reduction of infection rates and improve both animal and public health outcomes.
CONFLICT OF INTEREST
The authors declare that they have no financial or non-financial conflict of interest regarding authorship and publication of this article.
ACKNOWLEDGMENTS
The authors would like to thank the relevant shelter authorities across North Macedonia for granting permission to access the shelters and collect samples for this study, and the Faculty of Veterinary Medicine in Skopje for their support and collaboration.
AUTHORS’ CONTRIBUTION
AC conceived and designed the study. BC and AC wrote the manuscript, analyzed the data, and interpreted the results. BC, LR, JV, EAP, BX and IS performed the field and laboratory work. AC, JS and IC supervised the field and laboratory work and contributed to the interpretation of all results. AC conducted the statistical analysis and interpreted the results. All authors contributed to the final version of the manuscript.

 10.2478/macvetrev-2026-0012
10.2478/macvetrev-2026-0012




