Cisplatin is a widely used chemotherapeutic agent, but its clinical application is often limited due to its nephrotoxic effects, largely mediated through proinflammatory cytokines. This study investigates the comparative effectiveness of micellar and liposomal formulations of Coenzyme Q10 (CoQ10) in modulating serum levels of tumor necrosis factor-alpha (TNF-α) and interleukin-1 beta (IL-1β) in a rat model of cisplatin-induced nephrotoxicity. Ninety Wistar rats were divided into control group and five experimental groups receiving various treatments, including cisplatin alone, micellar CoQ10, liposomal CoQ10, and combinations thereof. Serum concentrations of TNF-α and IL-1β were measured at defined intervals using ELISA. The results demonstrated that both formulations of CoQ10 significantly reduced cytokine levels compared to cisplatin-only treated rats, with the liposomal form yielding superior anti-inflammatory effects. These findings suggest that liposomal CoQ10 may offer a more effective strategy for mitigating inflammation and nephrotoxicity induced by cisplatin.
INTRODUCTIONCisplatin is a first-line chemotherapeutic agent used in the treatment of various malignancies(
1). Despite its efficacy, its clinical application is limited due to significant side effects, particularly nephrotoxicity and myelosuppression (
2,
3). One of the primary mechanisms of cisplatin-induced nephrotoxicity is inflammation, driven by proinflammatory cytokines such as tumor necrosis factor-alpha (TNF-α) and interleukin-1 beta (IL-1β). These cytokines are released by damaged renal cells and promote the recruitment of inflammatory cells to the injured kidneys (
4,
5,
6). Elevated TNF-α levels have been observed as early as the first day following cisplatin administration (
7).
A key upstream regulator of TNF-α and IL-1β expression is nuclear factor kappa B (NF-κB), which also contributes to oxidative stress and the inflammatory cascade (
8). Cisplatin-induced renal damage is linked to mitochondrial dysfunction (
9), leading to increased production of reactive oxygen species (ROS) and release of pro-apoptotic factors, ultimately causing tubular cell death (
10).
Coenzyme Q10 (CoQ10), a lipid-soluble antioxidant molecule located in the inner mitochondrial membrane, has been shown to protect against nephrotoxicity (
11,
12) through its ability to reduce ROS and inflammation. Its micellar structure increases the protective effect against nephrotoxicity. Its anti-inflammatory effects are believed to be mediated by suppression of NF-κB activation (
13,
14). However, the therapeutic potential of CoQ10 is limited by its poor water solubility and low bioavailability.
Therefore, novel delivery systems such as liposomes have been explored. Liposomes, with their phospholipid bilayer structure, closely resemble cell membranes and offer improved stability, prolonged circulation time, and enhanced delivery of hydrophobic compounds like CoQ10 (
15,
16). These systems may enhance the biological activity of CoQ10 and improve its efficacy in mitigating cisplatin-induced nephrotoxicity.
Given these considerations, the present in vivo study aims to compare the anti-inflammatory effectiveness of liposomal versus micellar formulations of CoQ10 in rats with cisplatin- induced nephrotoxicity, by evaluating serum levels of TNF-α and IL-1β. In the same time, the results obtained in this study can confirm the expectations that both formulations of CoQ10 significantly reduce cytokine levels.
MATERIAL AND METHODS
Material and animals Cisplatin solution (1 mg/mL) was obtained from Accord, Latvia. Ascorbic acid and Tween 80 were purchased from Merck, Germany. Coenzyme Q10 (CoQ10) standard was acquired from Sigma-Aldrich, while thiopental sodium (500 mg) was procured from Ciron Drugs & Pharmaceuticals, India. The nanoliposomal formulations used in this study were previously formulated and optimized at the Faculty of Pharmacy, Skopje, with composition details available in Shalabalija et al. (
16). All other chemicals were of analytical or pharmaceutical grade and were used without further purification.
Male Wistar rats (250–300 g) were obtained from the animal facility of the Faculty of Natural Sciences and Mathematics, UKIM-Skopje. All procedures involving animals were conducted in accordance with the
Manual for the Care and Use of Laboratory Animals and approved by the Institutional Animal Ethics Committee of the University “Ss. Cyril and Methodius”-Skopje (No. 10-3196/1 from 20.09.2022) in line with European Commission Directive 86/609/EEC and CIOMS guidelines.
Preparation of CoQ10 micellar solution
A CoQ10 micellar solution (5 mg/mL) was prepared for intraperitoneal (i.p.) administration. Doses were adjusted based on the individual weight of each rat. CoQ10 powder was dissolved in 0.9% NaCl containing 1% Tween 80 and mixed continuously at 65 °C and 200 rpm for 15 min. Ascorbic acid (0.1% w/v) was added to preserve antioxidant stability.
Preparation of nanoliposomes with encapsulated CoQ10
Nanoliposomes (5 mg/mL CoQ10) were prepared using a modified dry lipid film hydration technique as described in Shalabalija et al. (
16). The resulting liposomes showed uniform size distribution (PDI: 0.26), an average diameter of 125 nm, and a sustained release of approximately 50% of the encapsulated CoQ10 within 24 h.
Animal grouping and treatment protocol
Ninety male normotensive Wistar rats were randomly divided into six groups (n=15 per group):
- Group 0 (control): received physiological saline with 1% Tween 80 and 0.1% ascorbic
- Group 1 (cisplatin): received a single dose of cisplatin (5 mg/kg, i.p.) on day 4.
- Group 2 (micellar CoQ10): treated daily with CoQ10 (10 mg/kg, i.p.) for 11 days.
- Group 3 (cisplatin+micellar CoQ10): received cisplatin (day 4) and daily micellar CoQ10 for 11 days.
- Group 4 (liposomal CoQ10): received nanoliposomal CoQ10 (10 mg/kg, p.) on days 1, 3, 5, 7, 9, and 11.
- Group 5 (cisplatin+liposomal CoQ10): received cisplatin (day 4) and liposomal CoQ10 on days 1, 3, 5, 7, 9, and 11.
Evaluation of TNF-α and IL-1β serum levels Blood samples were collected from the tail vein on days 0, 2, 6, and 12. Serum levels of TNF-α and IL-1β were quantified using ELISA kits (Sigma-Aldrich, RAB0476-1KT, RAB0273-1KT), following the manufacturer's instructions. The assay involved standard steps of antibody binding, washing, substrate reaction (TMB), and colorimetric measurement at 450 nm. The minimum detectable concentrations were 30 pg/mL for TNF-α and 0.3 pg/mL for IL-1β.
Statistical analysis
Statistical analyses were performed using Statistica 7.1 software (StatSoft Inc., USA). Descriptive statistics included mean, standard deviation, and 95% confidence intervals. Intergroup comparisons of TNF-α and IL-1β levels across treatment groups and time points were performed using one-way ANOVA followed by LSD post-hoc tests. A p-value of <0.001 was considered statistically significant.
RESULTSTNF-α serum concentration
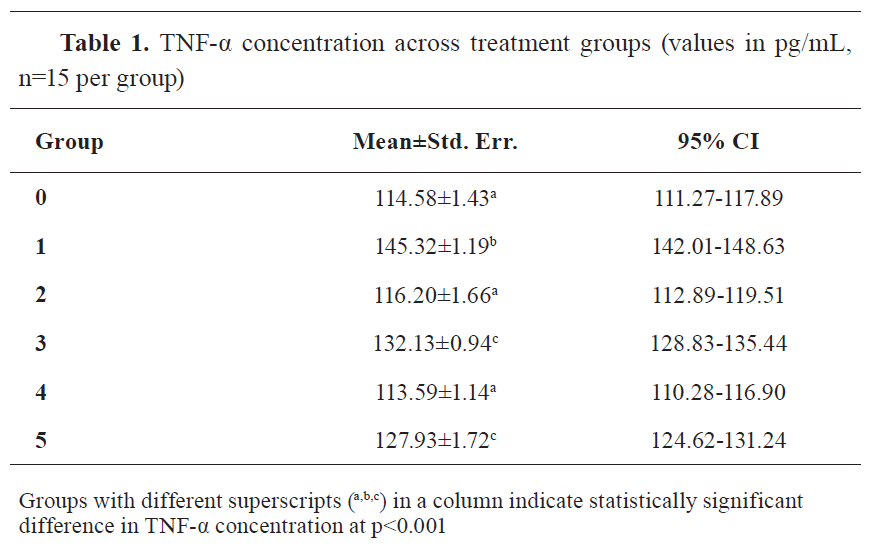
The results revealed significant differences in serum TNF-α levels among the six experimental groups (F=56.95; p<0.001). Group 1 (cisplatin only) exhibited the highest TNF-α concentration (145.32 pg/mL), which was significantly greater than that in the control group (114.58 pg/mL), as well as in all CoQ10-treated groups. The lowest TNF-α levels were recorded in Group 4 (liposomal CoQ10 alone; 113.59 pg/mL), which were even slightly lower than the control group, although this difference was not statistically significant (p=1.00). Groups 3 and 5, treated with micellar and liposomal CoQ10 in combination with cisplatin, respectively, also showed significant reductions in TNF-α compared to Group 1 (
Table 1).
Time-point measurements showed the highest TNF-α levels on day 2 (128.21 pg/mL), followed by gradual reductions by day 12 (121.02 pg/mL). The decrease from day 2 to day 12 was statistically significant (p<0.001), confirming the time- dependent inflammatory response and the modulation of cytokine levels by treatment.
IL-1β serum concentration
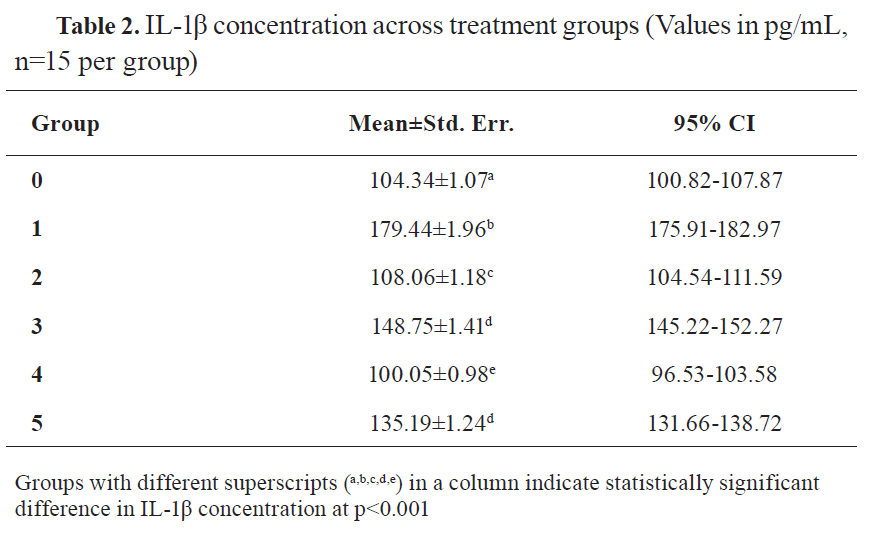
Significant differences in serum IL-1β levels were also observed among the groups (F=308.63; p<0.001). Group 1 again exhibited the highest concentration (179.44 pg/mL), while Group 4 (liposomal CoQ10 alone) demonstrated the lowest (100.05 pg/mL), significantly lower than all other groups including the control (104.34 pg/mL). Groups treated with cisplatin plus either micellar or liposomal CoQ10 (Groups 3 and 5) showed substantial reductions in IL-1β levels compared to cisplatin-only rats, with liposomal CoQ10 performing better (
Table 2).
Time-course data revealed no statistically significant changes in IL-1β levels over time (days 0, 2, 6, and 12), though day 6 showed a slight nonsignificant peak.
 DISCUSSION
DISCUSSION
This study demonstrates that both micellar and liposomal formulations of Coenzyme Q10 (CoQ10) significantly mitigate the cisplatin-induced increase in proinflammatory cytokines TNF-α and IL-1β in rats (
Table 1 and
Table 2). However, the liposomal formulation consistently showed superior effects in reducing these cytokines, suggesting improved bioavailability and efficacy compared to the micellar form.
The observed elevation in TNF-α and IL-1β in the cisplatin-only group aligns with prior studies indicating their key role in the pathophysiology of cisplatin-induced nephrotoxicity. These cytokines contribute to the recruitment of neutrophils and macrophages, generation of reactive oxygen species (ROS) (
17,
18) and direct tissue injury in the renal parenchyma (
19). NF-κB signaling, an upstream regulator of cytokine production, is known to be activated by cisplatin and serves as a target for anti-inflammatory agents.
CoQ10 exerts its effects by reducing NF-κB expression (
20) and ROS levels, and by stabilizing mitochondrial function. Similar studies outline the positive effects of CoQ10 supplementation in reducing the levels of TNF-α, IL-1β, and NF-κB expression (
11,
21,
22). However, its low solubility and rapid clearance hinder its clinical application. In this study, liposomal encapsulation of CoQ10 appears to overcome these limitations, leading to enhanced delivery and sustained release of the active compound (
9). This is evidenced by lower serum cytokine levels in animals treated with liposomal CoQ10 compared to those receiving the micellar formulation.
These findings are consistent with previous reports showing that nano-formulated CoQ10 improves pharmacokinetics, circulation time, and cellular uptake (
23). Liposomes, with their bilayer phospholipid membranes, mimic biological membranes, allowing for better interaction with cells and gradual intracellular release of CoQ10 (
24).
Although both formulations were effective, liposomal CoQ10 showed a greater reduction in TNF-α and IL-1β levels than micellar CoQ10, particularly when co-administered with cisplatin. This suggests that the liposomal system may provide better tissue distribution and prolonged anti-inflammatory action (
9).
The results support the potential use of liposomal CoQ10 as a protective agent in cisplatin-induced nephrotoxicity and possibly in other inflammatory conditions mediated by similar mechanisms.
CONCLUSIONCoenzyme Q10 emerges as a promising therapeutic agent for alleviating cisplatin-induced inflammation and nephrotoxicity. Both micellar and liposomal formulations demonstrated significant reductions in serum TNF-α and IL-1β levels, indicative of anti-inflammatory effects. However, the liposomal CoQ10 formulation exhibited superior efficacy, likely due to enhanced bioavailability, sustained release, and targeted delivery. These findings highlight the potential of liposomal drug delivery systems to improve the therapeutic impact of CoQ10 and support further investigation into its clinical application as a nephroprotective supplement during chemotherapy.
CONFLICT OF INTEREST
The authors declare that they have no financial or non-financial conflict of interest regarding authorship and publication of this article.
ACKNOWLEDGMENTS
The authors would like to express their gratitude to the technical staff and institutional support teams contributing to the animal care, laboratory analyses and statistical evaluation. No specific funding was received for this study.
AUTHORS’ CONTRIBUTION
All authors contributed substantially to the study design, experimental work, data analysis, and manuscript preparation. All authors have read and approved the final version of the manuscript.

 10.2478/macvetrev-2026-0013
10.2478/macvetrev-2026-0013

