Veterinary diagnostic techniques are pivotal for the early identification and effective treatment planning, making them central to advancing veterinary oncology. They underpin the accurate identification and characterisation of neoplastic diseases, helping to guide effective treatment planning and improve animal health outcomes. This review emphasises the critical role of diverse diagnostic techniques, including cytology, histopathology, immunohistochemistry, electron microscopy, radiographic imaging (X-ray, computed tomography, positron mission tomography, magnetic resonance imaging), serology, and molecular techniques such as polymerase chain reaction (PCR), fluorescence in situ hybridization, and gene sequencing, that are critical for tumour diagnosis in veterinary practice. Despite their indispensability, these methods face significant challenges, mainly a lack of comprehensive standardisation and limited validation of established protocols and grading schemes. Recognising these related issues will assist in resolving them, which is vital for improving diagnostic accuracy, promoting innovation, and equipping veterinary professionals to make informed and effective clinical oncology decisions, thereby advancing both veterinary care and research.
INTRODUCTION
Diagnostic techniques in veterinary clinical practice play a crucial role in identification and treatment planning, as well as in providing better care for various persistent health conditions (
1,
2). The same could be said for the pathological diagnosis of veterinary tumour cases, involving various techniques used in the identification and characterisation of these neoplasms (
1,
2). Without gainsaying, these techniques are invaluable but present their own challenges and limitations. Currently, there are only basic methodological standards for the appraisal of tumours in veterinary oncology practice, with only minimal efforts to validate published protocols and grading schemes (
1).
Studying these diagnostic techniques used in veterinary oncology and their associated problems (limitations and challenges) is important as they help to improve accuracy. They also encourage researchers and veterinary professionals to develop new methods or make better existing ones to improve on the accuracy and reliability. Furthermore, such studies foster innovation and advancements in the field. They also play an essential role in educating veterinary professionals and students about the principles, applications, and problems associated with diagnostic techniques to make informed decisions in clinical practice. Ultimately, this ensures appropriate veterinary care is delivered timely, generally benefitting animal health and welfare (
3,
4,
5).
Highlighted are the uses of each technique (cytology, histopathology, immunohistochemistry, transmission and scanning electron microscopy, radiographical pathology which involves the use of X-rays, computed tomography (CT scan), positron emission tomography (PET scans), magnetic resonance imaging (MRI), serology, molecular pathology techniques (polymerase chain reaction (PCR)), Fluorescence in situ hybridization (FISH), gene sequencing etc.), their associated problems and suggested solutions.
DIAGNOSTIC USES OF CYTOLOGY IN VETERINARY ONCOLOGY
Cytology is a simple, rapid, and inexpensive method used for diagnosis in veterinary practice by serving as a valuable diagnostic tool for the identification and characterisation of cancerous lesions in animals (
6,
7,
8). Veterinary cytology involves the microscopic examination of cells obtained through various non-invasive or minimally invasive methods, such as fine-needle aspiration (FNA) or impression smears (
8). Fine needle aspiration cytology (FNAC) is a comparatively simple, quick, and easily reproducible technique, which aids in pre-surgical diagnosis (
9,
10).
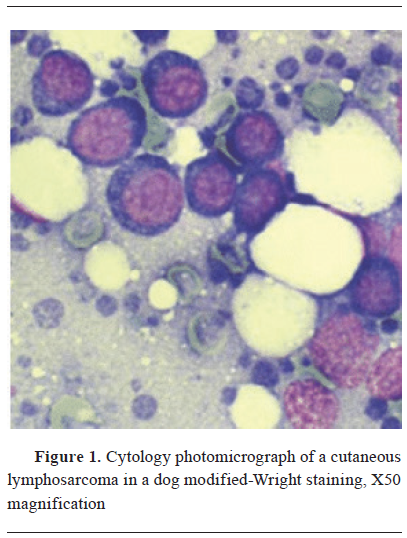
Cytology aids veterinarians in distinguishing between benign and malignant tumours (
Fig. 1), determining tumour types, and assessing the degree of malignancy (
2,
11,
12). The advantages of using cytology as a diagnostic modality in animals are abundant because the techniques employed for non-invasive collection methods lower the risk of complications, and the rapid availability of results facilitates patient evaluations and prompt clinical decisions (
11). Additionally, cytology is especially useful for monitoring cancer advancement and assessing response to therapy, enabling veterinarians to tailor treatment plans for individual cases (
13).
 Problems associated with the use of cytology as a diagnostic technique in veterinary oncology
Problems associated with the use of cytology as a diagnostic technique in veterinary oncology
Cytology relies on the examination of individual cells, and tumours may exhibit cellular heterogeneity, which could arise from complex genetic, epigenetic, and metabolic modifications and making it challenging to obtain a comprehensive representation of the entire tumour (
14,
15,
16). The presence of blood, inflammation, or normal cells in cytological samples can hinder the precise identification of neoplastic cells (
10,
17). In FNA samples, a small portion of the tumour, and sampling error can occur, leading to a misrepresentation of the tumour’s characteristics (
18,
19). In some cases, FNA may carry a risk of complications such as bleeding, infection, tumour seeding or the possibility of tumour cell dissemination as has been evidenced in human oncology practice (
20,
21).
DIAGNOSTIC USES OF HISTOPATHOLOGY IN VETERINARY ONCOLOGYHistopathology offers greater diagnostic precision compared to cytology for identifying tumor type. It is crucial for differentiating metastatic tumors from primary benign or preneoplastic lesions (
Fig. 2a-d), evaluating prognosis, and guiding treatment strategies. The functions that imaging modalities and tumor markers alone cannot fully replace (
22,
23,
24). Histopathology is a fundamental diagnostic tool in veterinary oncology and is considered as a gold standard in tumour diagnosis. It provides crucial insights into the microscopic structure of tissues (identifying specific cell types, irregularities of cell shapes, tissue distributions patterns, and characteristics) and aids in the identification and characterisation of tumours in animals, deciding if the parts of the tissue are cancerous, and determining the metastatic grade and stage thus making tumour diagnosis and also classifying such tumours (
1,
24,
25). Examining tissue samples under a microscope allows pathologists to facilitate accurate categorisation of different types of neoplasms prevalent in veterinary patients (
24,
26). By assessing cellular abnormalities, mitotic activity, and invasion into surrounding tissues, veterinarians can gauge the cancer’s aggressiveness and predict its potential for metastasis, which are crucial factors in identifying prognostic and therapeutic targets (
25,
27,
28).
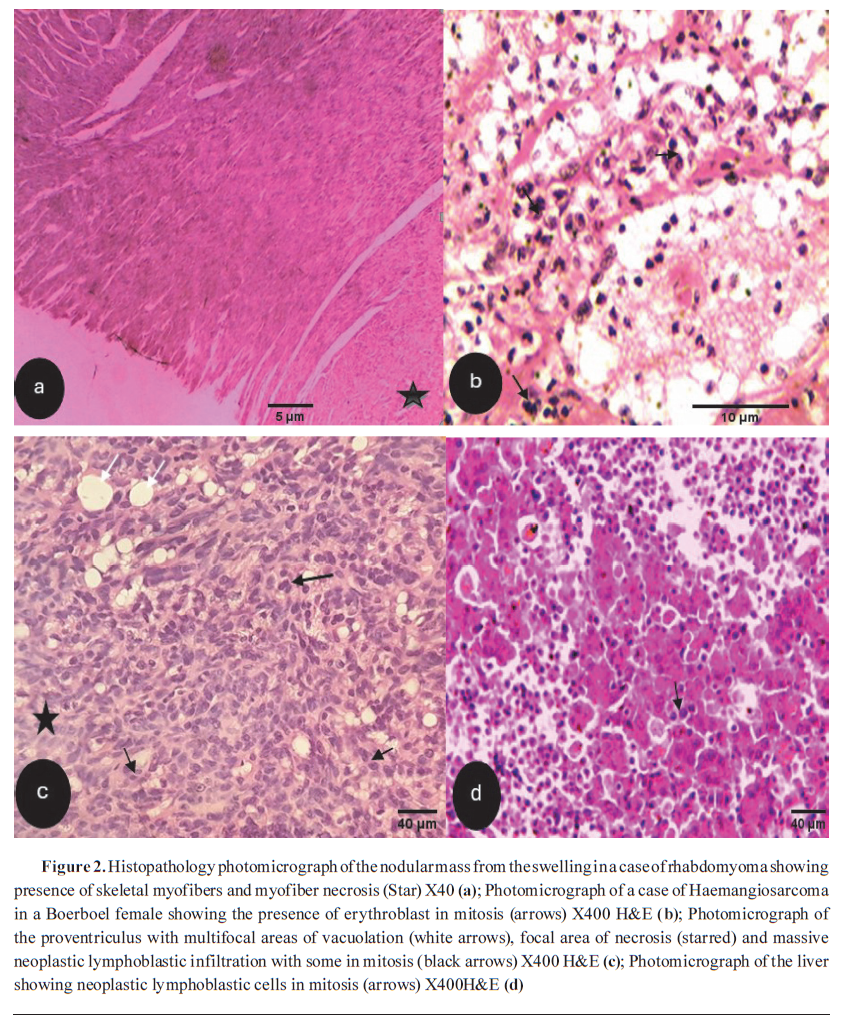
Histopathology is also very instrumental in evaluating treatment response. Periodic examination of post-treatment tissue samples enables veterinarians to assess the effectiveness of interventions, guiding adjustments to treatment plans and ensuring the best possible outcomes for the animal (
1). Furthermore, histopathology contributes to research efforts in veterinary oncology. The detailed examination of tissues helps in understanding the underlying molecular and cellular mechanisms of cancer, paving the way for the development of novel diagnostic methods and targeted therapies (
1,
29).
Therefore, histopathology is an indispensable diagnostic tool in veterinary oncology. Its applications span tumour diagnosis, classification, grading, staging, differentiation between benign and malignant tumours, evaluation of treatment response, and contributions to cancer research. As veterinary medicine advances, histopathology remains a cornerstone, providing accurate and essential information for optimal cancer management in animals.
Problems associated with the use of histopathology as a diagnostic technique in veterinary oncology
Despite its pivotal role, histopathology faces challenges, including variability in interpretation and potential sampling errors. Efforts to standardise protocols and enhance training are ongoing to improve the consistency and reliability of histopathological diagnosis of neoplasms (
30,
31). Histopathological assessment of oncology cases is usually variable and subjective because it requires in-depth clinical and pathological knowledge of these cases, but also is majorly dependent on skill and experience, and most time requires a second opinion (
24). Other associated problems include: the accuracy of histopathology heavily depends on the quality and representativeness of the tissue sample (
32). Inadequate or biased sampling can lead to misinterpretation of the tumour type and grade, and improper tissue fixation and processing can introduce artefacts that may affect the accuracy of histopathological interpretation (
1,
32).
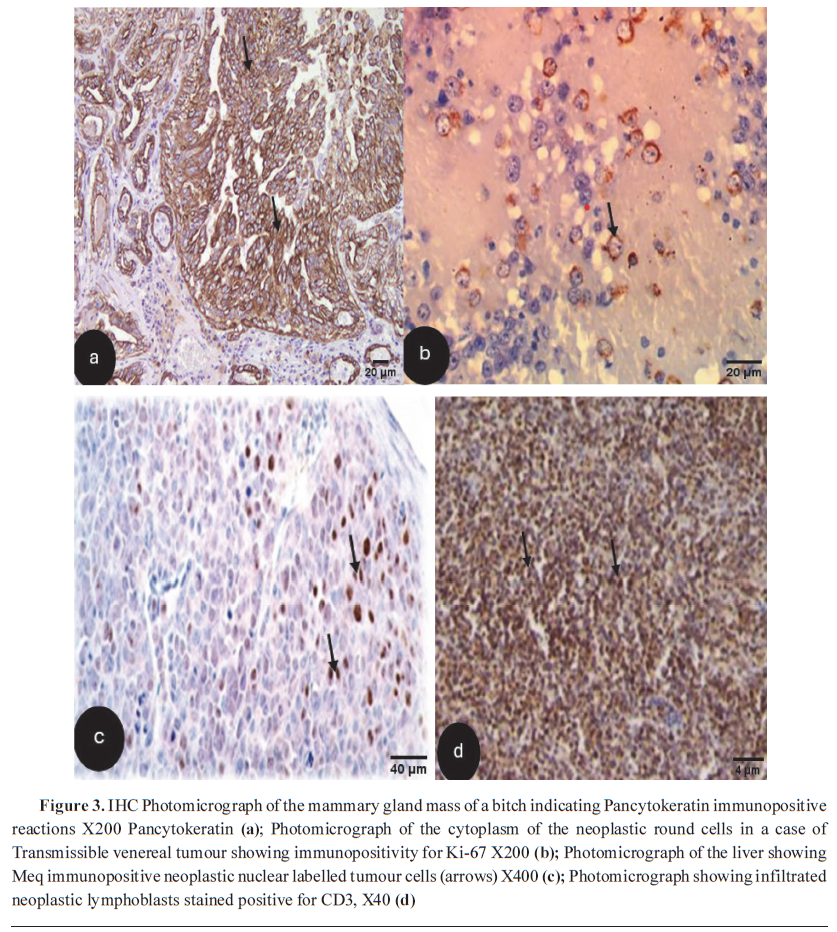
DIAGNOSTIC USES OF IMMUNOHISTOCHEMISTRY (IHC) IN VETERINARY ONCOLOGYImmunohistochemistry (IHC) is a vital diagnostic technique in veterinary oncology that bridges three disciplines: immunology, histology, and chemistry, offering valuable insights and enabling antigen recognition in histological sections by specific antibodies. The antigen–antibody binding is then visualised using light or fluorescent microscopy as a coloured histochemical reaction. This method involves employing specific antibodies to detect, express, and visualise target proteins within the context of spatial localisation and tissue morphology, thus aiding in the identification and characterisation of various cancers in animals (
Fig. 3a and
3b) (
33,
34). Consequently, by detecting the expression of tumour-specific antigens, IHC tumour diagnosis is made (
35). The importance of IHC in veterinary oncology research has grown due to increased sensitivity and specificity, better identification of various antigens within a single histological tissue section, concurrent assessment of multiple samples through tissue microarrays, advanced antigen retrieval techniques, and enhanced automation (
33,
35).
IHC has become a routine supplement to the classic morphologic approach of investigational pathology (
36). A key diagnostic use of immunohistochemistry (IHC) in veterinary oncology is identifying and classifying the origin of tumours. IHC plays a pivotal role in predicting the behaviour of tumours. Though its main application in veterinary oncology is in the characterisation and diagnosis of neoplastic diseases, current trends highlight a central role for IHC in prognosis and theranostics (
36,
37). In veterinary oncology, immunohistochemistry (IHC) is utilised for identifying specific biomarkers associated with neoplasms. This aids in early diagnosis and monitoring of disease progression, contributing to timely interventions and improved patient outcomes (
38). IHC is instrumental in guiding targeted therapies. The identification of specific protein targets allows for the development of targeted treatments tailored to the unique molecular characteristics of individual tumours, enhancing the precision and efficacy of therapeutic interventions (
39).
In summary, IHC is a cornerstone diagnostic technique in veterinary oncology. Its applications span tumour classification, prediction of behaviour, guiding targeted therapies, early detection, and differentiation between benign and malignant tumours. As veterinary medicine continues to advance, IHC remains a valuable tool in enhancing the precision, accuracy, and individualised care provided to animals with cancer (
1).
 Problems associated with the use of immunohistochemistry as a diagnostic technique in veterinary oncology
Problems associated with the use of immunohistochemistry as a diagnostic technique in veterinary oncology
Immunohistochemistry (IHC), although a valuablediagnostictechniqueinveterinaryoncology, aids in the identification and characterisation of specific proteins within tissues. Its application is not without challenges and limitations. The availability of specific antibodies for the diversity of veterinary species may be limited, restricting the range of markers that can be used for immunohistochemical analysis. Variability in staining protocols, antibody specificity, and tissue handling can lead to inaccurate interpretations, impacting the reliability of the diagnostic information provided (
34,
40). The standardisation of IHC procedures is essential to ensure consistency across various laboratories and practitioners. However, it is often lacking in most veterinary oncology diagnostic laboratories (
1,
41). The cost associated with IHC procedures, including the acquisition of specialised antibodies and equipment, can be a limiting factor. This financial burden may impact the accessibility of IHC as a diagnostic tool for certain veterinary clinics and pet owners, potentially influencing decision-making in tumour characterisation (
42).
Interpretation of IHC results requires expertise. The possibility of variability in interpretation among pathologists can affect the consistency of diagnoses, emphasising the need for standardised guidelines to enhance diagnostic reliability (
43). There have been no standardised guidelines for use in veterinary oncology practice until the work by Priest et al. (
44). This could lead to subjective assessments (
43). Another significant problem is the potential for false positives or false negatives in IHC results. The availability of specific and validated antibodies for veterinary species is another challenge. Limited availability or cross- reactivity issues with antibodies designed for human tissues can compromise the accuracy of IHC analyses in animals (
45,
46). Developing and validating species-specific antibodies is key for consistent and accurate results.
Proper storage and handling of tissue samples are important for effective IHC analysis. Inadequate fixation, processing, or storage conditions can cause protein degradation, potentially impacting the quality of IHC results. Standardised protocols for tissue handling have become essential to ensure optimal staining outcomes (
43,
47). Immunohistochemistry is an important diagnostic tool in veterinary oncology. However, several challenges must be addressed, including issues of standardisation, availability of antibodies, variability in interpretation, cost constraints, tumour heterogeneity, and complexities in sample handling. Efforts toward standardising protocols, developing species-specific antibodies, and enhancing training for consistent interpretation are essential to maximise the utility of IHC in the accurate diagnosis and characterisation of cancer in veterinary patients.
DIAGNOSTIC USES OF ELECTRON MICROSCOPY IN VETERINARY ONCOLOGY
Since its development in 1931, transmission electron microscopy (TEM) has significantly advanced medical research. By 1953, ultramicroscopic details of animal and tumour cells were published (
48). TEM is used to analyse subcellular structures and subtle changes associated with various pathological conditions, including neoplastic diseases. Alongside scanning electron microscopy (SEM), TEM is a valuable research tool for studying subcellular spaces in detail (
48).
Therefore, both TEM and SEM are powerful diagnostic tools in veterinary oncology, offering detailed insights into the microscopic structure of neoplastic cells and tissues (
49). Scanning electron microscopy (SEM) is used in veterinary oncology to study cancer cell surface characteristics, aiding in tumour classification and providing important information for diagnosis (
50). In contrast, transmission electron microscopy (TEM) offers cross-sectional views of cellular structures, facilitating the visualisation of intracellular organelles, nuclear morphology, and other ultrastructural details. This is vital for identifying specific cancer types and understanding their biological behaviour (
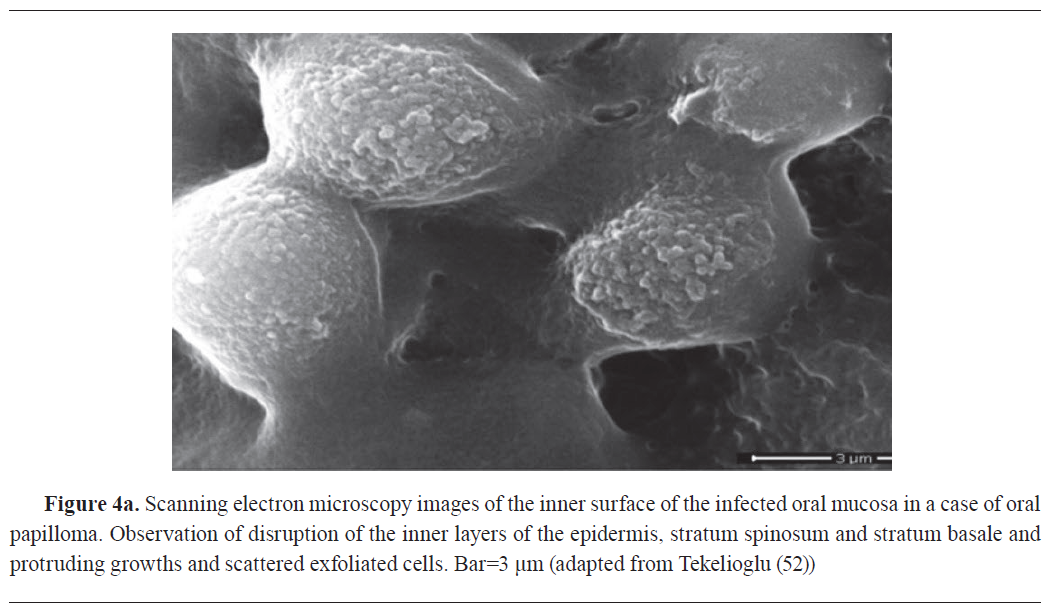
51). SEM and TEM are used in veterinary oncology to identify cellular abnormalities, including changes in cell shape, size, and organelle distribution (
50,
51). These techniques are particularly valuable for studying rare or poorly characterised neoplasms, providing essential information for proper diagnosis and treatment planning (
Fig. 4a &
b).
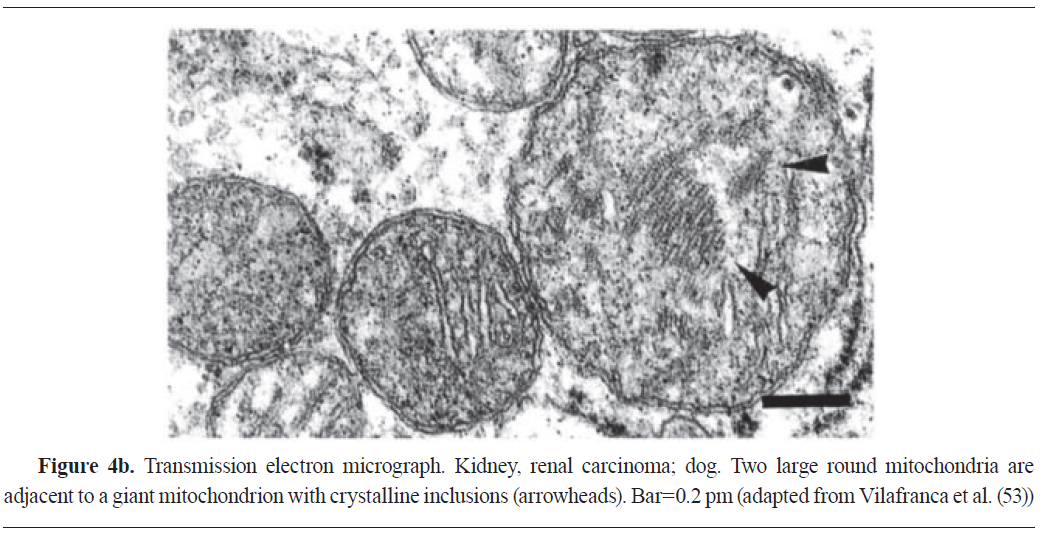
Furthermore, SEM and TEM contribute to cancer research by facilitating detailed investigations into the ultrastructural changes associated with tumour development and progression (
48,
50). Microscopic techniques facilitate a comprehensive understanding of veterinary cancers at both cellular and molecular levels, thereby contributing to the development of targeted therapies and personalised treatment plans (
48,
50). SEM and TEM are indispensable tools in veterinary oncology, offering detailed imaging of cancer cell morphology and ultrastructure. Their diagnostic applications encompass tumour classification, identification of cellular abnormalities, and advancing research to improve our understanding of cancer biology. These electron microscopy techniques significantly contribute to the precision and depth of veterinary cancer diagnostics, ultimately enhancing the management and care of animals with cancer.
Problems associated with the use of electron microscopy as a diagnostic technique in veterinary oncology
Electron microscopy in veterinary oncology faces challenges primarily related to the intricate and time-consuming sample preparation process. This labour-intensive process may introduce artifacts, affecting result accuracy. The high costs associated with acquiring and maintaining electron microscopy equipment present a financial hurdle, limiting its accessibility for veterinary clinics and research institutions. This impediment prevents its widespread adoption as a routine diagnostic tool (
42,
54). The size limitations of electron microscopy machines further constrain their utility. The small sample chamber size may limit imaging for larger tissue specimens or those with specific anatomical considerations, reducing its applicability across various cases (
55,
56). While electron microscopy provides high resolution, it does not offer real- time imaging capabilities, unlike other modalities that can capture dynamic tissue processes. This limitation underscores the necessity for a complementary approach in veterinary oncology diagnostics (
56).
Interpreting electron microscopy images requires specialised skills, and the complexity of ultrastructural analysis needs highly trained experts. A shortage of skilled microscopists may cause inconsistencies in interpretation and diagnosis (
57). Moreover, electron microscopy may not be universally suitable for all tumour types or cases, as certain tumours may lack distinctive ultrastructural features. Therefore, it is often used alongside other diagnostic methods for a comprehensive assessment (
48,
58). Ongoing efforts to streamline processes, enhance accessibility, and integrate electron microscopy judiciously within diagnostic frameworks are essential for maximising its benefits in precise cancer characterisation for veterinary patients.
DIAGNOSTIC USES OF RADIOGRAPHY IN VETERINARY ONCOLOGYRadiography is still the predominant first-line imaging procedure for many queries in veterinary oncology. It is often used as a comparatively inexpensive and easily accessible screening test. Radiography uses electromagnetic x-rays to visualise internal body structures based on variations in their opacity (
59). The major advantages of radiographs are the comparatively low cost, the easy global depiction of large body parts (in small animals), and the excellent bone imaging capacity (
59).
Radiography is crucial in veterinary oncology, helping detect, stage, and monitor animal tumours. It allows veterinarians to see tumour size, location, and metastasis, aiding in biopsy and treatment planning (
60,
61). Furthermore, X-rays are instrumental in monitoring treatment efficacy, identifying complications, and providing ongoing surveillance for tumour recurrence. This ultimately enhances the quality of care and outcomes for cancer patients within veterinary practice (
62).
Problems associated with the use of radiography as a diagnostic technique in veterinary oncology
Radiography is a widely used diagnostic technique in veterinary oncology; however, it has certain challenges and limitations. A major one is the limited soft tissue contrast provided by traditional radiography, making it challenging to distinguish between different types of tumours (
60,
63,
64). The possibility of superimposition of overlying structures is also a disadvantage (
59). The exposure to ionizing radiation during radiography poses concerns, especially for animals undergoing repeated imaging or those with pre- existing health issues (
65,
66). Size limitations of standard radiography equipment can be a challenge, particularly for larger animals or those with unique anatomies. The restricted field of view may hinder the comprehensive assessment of tumours, impacting the ability to visualize the full extent of the disease or its relationship with surrounding structures (
60,
62,
65).
Additionally, the differentiation between benign and malignant tumours is almost impossible using radiography, as certain tumours may exhibit similar appearances. Thus, this limitation underscores the importance of combining radiography with other imaging modalities and diagnostic techniques, most importantly cytology and histopathology for a more comprehensive evaluation (
67). Interpretation of radiographic images requires expertise, and variations in interpretation among different practitioners can impact diagnostic accuracy. Standardisation of protocols and continuous training are essential to ensure consistent and reliable interpretations (
68,
69). While radiography is a valuable diagnostic tool in veterinary oncology, challenges such as limited soft tissue contrast, radiation exposure concerns, size limitations, lack of three-dimensional information, and potential difficulties in tumour differentiation need to be considered. Combining radiography with complementary imaging techniques and maintaining a balance between diagnostic efficacy and minimizing risks contribute to its effective use in the diagnosis and management of cancer in veterinary patients.
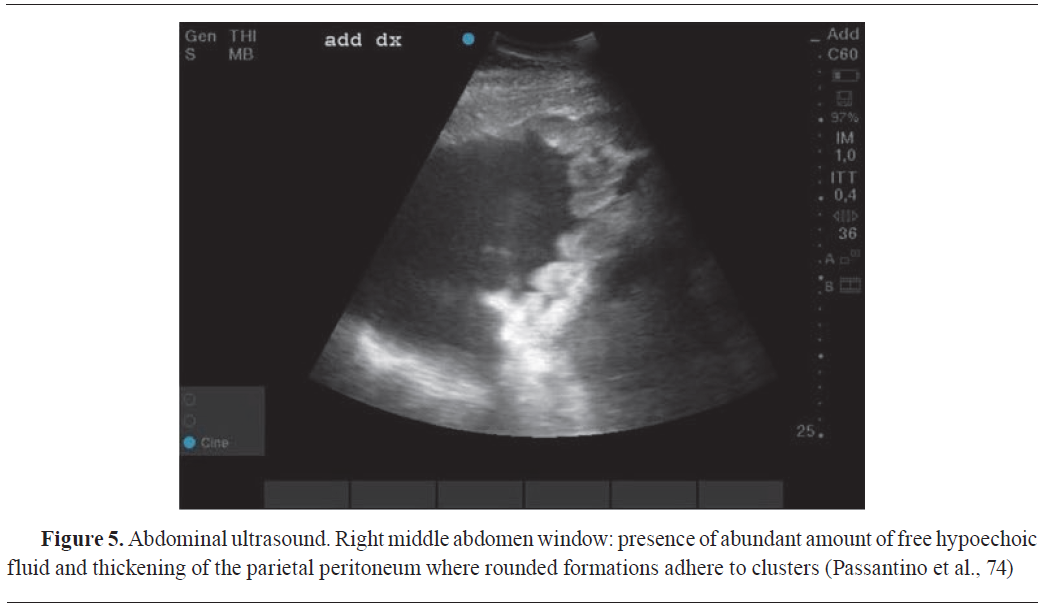
DIAGNOSTIC USES OF ULTRASONOGRAPHY IN VETERINARY ONCOLOGYDiagnostic ultrasound utilizes ultrasound waves to make image of internal organs tumours (
Fig. 5). It is based on the reflection of sound waves from the border between two tissues with different acoustic impedances. The acoustic impedance of targeted tissues depends on its physical density and the achievable velocity of the sound waves sent from the transducer into the tissue (
59,
61). In veterinary clinical practice, contrast-enhanced ultrasonography allowed the precise assessment of visceral organ vascularization in healthy dogs and the differentiation between malignant and benign visceral organ nodules in dogs and cats based on perfusion patterns (
Fig. 5) (
70). Compared with the other diagnostic imaging techniques, ultrasonography has the advantages of fast real-time imaging, low cost, and the lack of x-ray exposure. Disadvantages are limited imaging of the bone and air-filled spaces, comparably low image resolution, and a limited body penetration (
59,
61).
On the other hand, elastography is a relatively novel ultrasound method that, alongside the previously mentioned contrast-enhanced ultrasonography technology (
71), provides non- invasive diagnostic data based on the stiffness of tissues, evaluating their stiffness or elasticity (
72). The elastography results obtained from various types of elastography (static and dynamic elastography) can aid in the identification and differentiation between malignant and benign tissues (
72,
73).
 Problems associated with use of ultrasonography as a diagnostic technique in veterinary oncology
Problems associated with use of ultrasonography as a diagnostic technique in veterinary oncology
Ultrasonography is a valuable diagnostic technique in veterinary oncology; however, it is not without challenges and limitations. In certain cases, overlapping tissues in diagnostic images may hinder the accurate localization and characterization of tumors. Imaging techniques may not provide sufficient resolution to distinguish between different tumour types or to assess the extent of invasion into surrounding tissues (
75,
76). The quality of ultrasonographic images can vary based on the skill and experience of the operator, potentially impacting the accuracy of tumour detection and characterisation (
77).
Ultrasonography may encounter difficulties in delivering detailed soft tissue contrast, which can reduce its effectiveness in differentiating between various types of tumours based on their tissue characteristics. Additionally, the inability to obtain three-dimensional images limits the depth of information available for certain complex cases (
75,
78). While ultrasound is generally considered a non-invasive technique, obtaining high-quality images may require patients to be still, necessitating sedation or anaesthesia. This introduces potential risks, especially in older or compromised patients, and may affect the overall health assessment of the animal (
79,
80). The lack of standardised protocols for veterinary ultrasound may result in variations in image acquisition and interpretation, impacting the consistency of diagnoses across different practitioners. Interpretation of ultrasound images requires specialized expertise, and the availability of trained veterinary sonographers may be limited, leading to variations in diagnostic accuracy (
79,
81).
Furthermore, ultrasound’s efficacy in assessing certain tumour characteristics, such as vascularity, may be limited, potentially hindering the comprehensive understanding of tumour biology (
76). The potential for overdiagnosis or false positives must be considered, as ultrasound may detect lesions that are clinically insignificant, leading to unnecessary interventions and stress for pet owners (
2,
82).
Like contrast-enhanced ultrasonography technology, elastography encounters specific challenges in veterinary oncology due to the lack of standardized protocols, leading to discrepancies in both application and interpretation (
83,
84). Significant anatomical and physiological variations among different species and breeds make it difficult to establish normal tissue stiffness ranges (
83,
85). Furthermore, factors such as high costs, limited access to elastography equipment, and ethical and logistical hurdles in veterinary clinical practise hinder the routine clinical use of elastography in veterinary oncology. Additionally, animal movement during imaging may introduce artifacts, while sedation -which is used to minimize motion-might influence tissue elasticity and the resulting diagnosis (
42,
86).
Despite these challenges, ultrasonography remains a valuable and widely used imaging modality in veterinary oncology, contributing to the overall management and care of animals with cancer.
DIAGNOSTIC USES OF COMPUTED TOMOGRAPHY (CT) IN VETERINARY ONCOLOGY
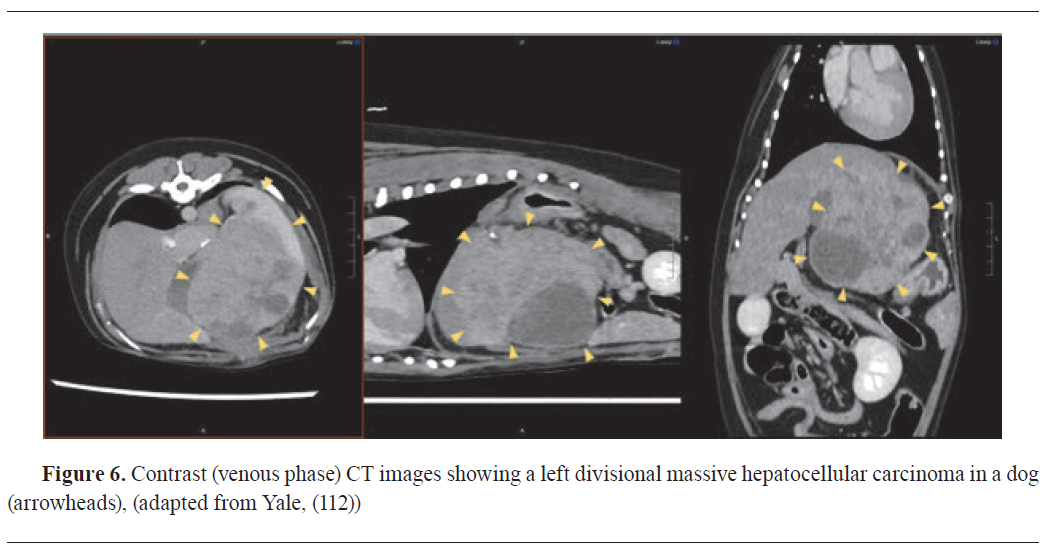
Computed tomography (CT) has emerged as an indispensable tool in veterinary oncologic practice, offering precise tumour staging and dependable evaluation of chemotherapeutic response during follow-ups. Increasingly, CT plays a pivotal role in preoperative planning, enabling surgeons to evaluate tumour size and determine the feasibility of complete resection (
Fig. 6) (
87,
88). CT imaging also plays a crucial role in radiotherapy treatment planning (
63,
89). The strength of CT scanning lies in its high spatial resolution, allowing for anatomical characterization of neoplasms (
63,
90).
Contrast-enhanced computed tomography (CECT) provides a valuable tool in veterinary oncology for detecting cancer-associated angiogenesis through the visualization and measurement of tumour blood vessels (
91,
92). Given that angiogenesis affects contrast enhancement in computed tomography (CT), CECT offers indicators for tumour angiogenesis (
92). Dynamic contrast-enhanced CT (DCECT) has been utilized in evaluating blood flow and volume in oronasal tumours in dogs, showing variations that could influence prognosis and treatment strategies (
93). Although there are some restrictions, the integration of contrast imaging with CT improves the capacity to diagnose, track tumour development and treatment response in veterinary patients (
94,
95).
Problems associated with use of computed tomography as a diagnostic technique in veterinary oncology
Computed tomography (CT) is a valuable diagnostic tool in veterinary oncology, but its use is not without challenges and limitations (
52,
88). One significant problem is the cost associated with CT procedures, including equipment acquisition and maintenance, potentially limiting accessibility for veterinary clinics and pet owners, thus leading to potential delays in diagnosis and treatment. The need for anesthesia during CT scans poses risks, especially for older or compromised patients, impacting overall health assessments (
42,
88).
Additionally, the lack of standardized protocols for veterinary CT may result in variations in image quality and interpretation. Image artifacts, such as motion artifacts and metal artifacts, can compromise diagnostic accuracy, affecting the precision of tumour localization and characterisation (
96,
97). Size limitations of CT machines may hinder imaging for larger animals or those with specific anatomical considerations. The relatively high radiation exposure associated with CT scans raises concerns about cumulative radiation effects, especially with repeated imaging over time (
98,
99).
CT’s inability to provide detailed soft tissue contrast may limit its effectiveness in certain cases, particularly for distinguishing between different types of tumours (
100). Interpretation of CT images demands specialized expertise, and limited availability of veterinary radiologists may lead to variations in diagnostic accuracy (
64,
101). Contrast agent administration, although helpful, may be contraindicated for animals with renal or hepatic issues, potentially restricting its use in certain cases. Furthermore, there is a potential for overdiagnosis or false positives, as CT may identify lesions that are clinically insignificant, leading to unnecessary interventions and stress for pet owners (
102).
The challenges associated with the use of CT in veterinary oncology underscore the need for ongoing research and technological advancements. Addressing issues such as cost, availability, standardized protocols, and specialized training for veterinary professionals can enhance the utility of CT as a diagnostic tool for animal cancer patients. Despite these challenges, CT remains a valuable technology in veterinary oncology, contributing to improved patient care and outcomes.
DIAGNOSTIC USES OF POSITRON EMISSION TOMOGRAPHY (PET) IN VETERINARY ONCOLOGY
Positron emission tomography (PET) plays a critical role in the diagnosis, staging, treatment planning, and therapeutic monitoring of neoplastic diseases. PET imaging complements computed tomography (CT) and magnetic resonance imaging (MRI) (
64,
88). The PET imaging offers functional insights, mostly through assessment of metabolic activity (
64,
88). For image diagnosis in cancer cases, the most widely used radiotracer, (18) F-fluorodeoxyglucose (F-18 FDG), a glucose analog, highlights regions of increased glycolytic activity, a hallmark of many malignancies (
88). CT images provide excellent anatomic depiction and aid in interpretation of the PET findings (
87,
88). PET/CT fusion imaging, commercially introduced in 2001, has since evolved through significant technological advancements, enhancing anatomical localization and diagnostic accuracy. Beyond F-18 FDG, novel tracers such as Radiolabelled tracers such as (
18) F-fluorothymidine (FLT) allow PET’s utility by enabling visualization of cellular proliferation (
103,
104). Although, traditionally employed in human oncology, PET imaging is now increasingly adopted in veterinary oncology as well (
61,
87).
Problems associated with use of positron emission tomography as a diagnostic technique in veterinary oncology
Positron emission tomography (PET) is a sophisticated diagnostic technique in veterinary oncology, but it comes with certain challenges. One significant problem is the limited availability of PET facilities for veterinary patients, restricting access and leading to delays in diagnosis and treatment planning (
42,
105). Additionally, the high cost of PET scans poses financial challenges for both veterinary clinics and pet owners, potentially influencing the decision to utilize this advanced imaging modality (
42). The use of radioactive tracers in PET scans raises safety concerns, as handling and disposal of radioactive materials require specialized protocols. This aspect may pose logistical challenges and increase the overall complexity of the diagnostic process (
88,
106). PET’s relatively lower spatial resolution compared to other imaging techniques may limit its ability to precisely localize small lesions or assess fine anatomical details. This limitation can impact the accuracy of tumour localization and characterisation, particularly in cases with subtle or small lesions (
107). PET’s sensitivity to motion artifacts presents challenges in imaging animals, as patient movement during scanning may compromise image quality. This issue is particularly relevant in veterinary patients who may have difficulty remaining still during the procedure (
88,
105).
Specialized training is required for the interpretation of PET images, and there may be variations in expertise among interpreting clinicians. Standardisation of interpretation criteria and ongoing training are essential for ensuring consistent and accurate diagnostic assessments (88, 105). While PET is excellent for detecting metabolic activity in tissues, it may not always distinguish between benign and malignant lesions solely based on metabolic patterns. This limitation emphasizes the need for combining PET with other diagnostic methods for a comprehensive assessment (
105). PET’s dependence on specialized facilities and equipment may limit its use in smaller veterinary clinics or regions with limited resources. This constraint underscores the importance of developing more accessible and cost-effective alternatives for comprehensive cancer imaging in animals (
42,
106). Additionally, the use of anesthesia in veterinary patients undergoing PET scans introduces potential risks, especially in older or compromised individuals. Anaesthesia considerations add a layer of complexity to the overall health assessment of the animal (
108,
109).
In conclusion, while positron emission tomography is a valuable tool in veterinary oncology, challenges such as limited availability, high costs, safety concerns with radioactive tracers, lower spatial resolution, sensitivity to motion artifacts, interpretation expertise, and the need for specialized facilities must be addressed. Ongoing advancements and efforts to enhance accessibility, standardize procedures, and broaden expertise will contribute to maximizing the benefits of PET in the precise diagnosis and management of cancer in veterinary patients.
DIAGNOSTIC USES OF MAGNETIC RESONANCE IMAGING (MRI) IN VETERINARY ONCOLOGY
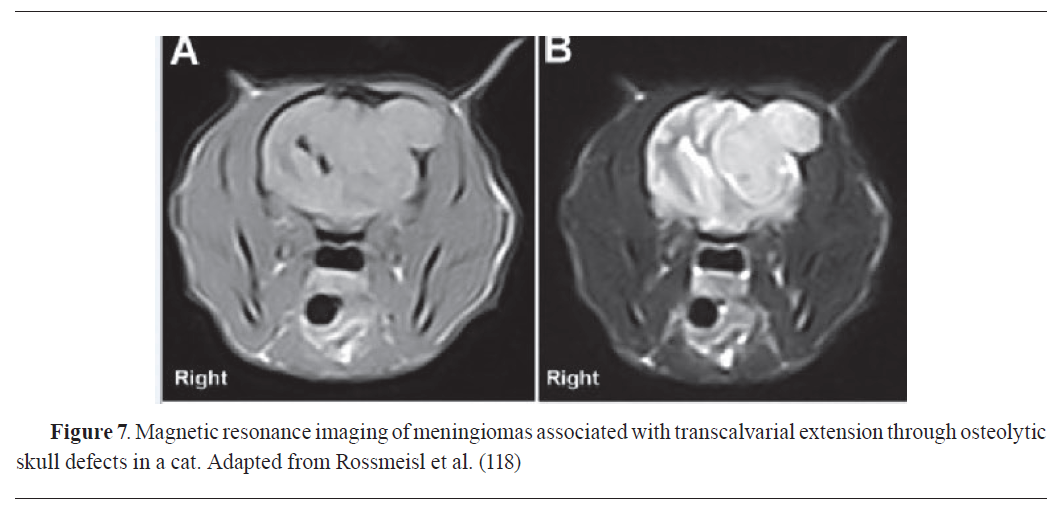
Magnetic resonance imaging (MRI) is a vital diagnostic tool in veterinary oncology, offering detailed images of soft tissues, organs, and tumours (
Fig. 7). MRI has been with great success in the diagnosis of tumours in animals (
110,
111). MRI is a good diagnostic imaging modality to detect neoplastic lesions and to diagnose tumour types in dogs by precise tumour localization, aiding in identifying the exact location and extent of neoplastic growth (
Fig. 7) (
61,
110,
111). Whole animal MRI can be performed on dedicated high-field MRI and nuclear magnetic resonance (NMR) scanners (
61,
112,
113). Magnetic resonance Imaging is used in detection of malignancy, staging, monitoring of therapy response, biopsy guidance etc (
61,
87,
100).
MRI provides excellent soft tissue contrast for tumour characterisation, helping determine the tumour’s nature and potential involvement of surrounding tissues. In tumour staging, MRI assists veterinarians in assessing tumour size, invasion into neighbouring structures, and potential metastatic spread (
61,
114). Contrast-enhanced MRI provides insights into tumour vascularization, evaluating blood supply and contributing to the understanding of tumour biology. Serial MRI scans are employed for assessing treatment responses, indicating changes in tumour size and characteristics in response to chemotherapy or radiation therapy (
115,
116). Regular MRI examinations allow longitudinal monitoring of tumour growth, aiding veterinarians in treatment decision-making and intervention timing. In preoperative planning, MRI offers detailed anatomical information, helping surgeons plan optimal approaches for tumour removal while minimizing damage to healthy tissues (
61,
117).
Moreover, MRI identifies cystic components within tumours, providing additional information about tumour composition and aiding in characterizing certain types of neoplasms. Lastly, MRI is extensively used in research studies and clinical trials in veterinary oncology, contributing to the development of innovative diagnostic and treatment strategies for cancer in animals (
117).
Problems associated with use of magnetic resonance imaging as a diagnostic technique in veterinary oncology
The use of magnetic resonance imaging (MRI) in veterinary oncology faces several challenges and limitations. The significant associated cost with MRI procedures, including acquiring and maintaining equipment, may limit access for both veterinary clinics and pet owners, potentially influencing decisions on its use in veterinary oncology (
42,
119,
120). Anaesthesia is often required during MRI examinations to ensure animals remain still for accurate imaging, introducing potential risks, especially in older or compromised patients, and impacting overall health assessments (
108,
109).
Additionally, the lack of standardized protocols for veterinary MRI may result in variations in image quality and interpretation. Image artifacts, such as motion and susceptibility artifacts, can compromise diagnostic accuracy, affecting the precision of tumour localization and characterisation (
121). Size limitations of MRI machines may hinder imaging for large or exotic animals, and the relatively long imaging times associated with MRI may be impractical for certain animals, particularly those struggling to remain still (
121). MRI’s sensitivity to metallic objects poses challenges, requiring careful evaluation before conducting scans on patients with metal implants or foreign bodies (
122).
Interpreting MRI images in veterinary oncology demands specialized expertise, and limited availability of veterinary radiologists may lead to variations in diagnostic accuracy based on the interpreting clinician’s experience. Contrast agent administration, though helpful, may be contraindicated for animals with renal or hepatic issues, potentially restricting its use in certain cases (
55,
119). Moreover, the potential for overdiagnosis or false positives must be considered, as MRI may identify lesions that are clinically insignificant or non-progressing, leading to unnecessary interventions and stress for pet owners. In addition, tumours and tumour-like lesions of the central nervous system especially in the brain usually look alike on ultrasound, CT or MRI studies thus, differentiating brain tumors from non-neoplastic lesions in veterinary clinical is challenging, leading to diagnostic uncertainties and errors (
123,
124). Cerebrospinal fluid analysis can support the diagnosis, with tumors showing elevated protein and occasional neoplastic cells (
15).
The challenges underscore the need for ongoing research and technological advancements, focusing on issues such as cost, availability, standardized protocols, and specialized training for veterinary professionals. Despite these challenges, MRI remains a valuable and evolving technology in veterinary oncology, contributing to improved patient care and outcomes. Recent advances in quantitative imaging such as advanced MRI techniques like perfusion- weighted imaging (PWI) and diffusion-weighted imaging (DWI), and MR spectroscopy provide additional clues regarding evaluating tumour extent, predicting grade, and assessing treatment response, together with artificial intelligence are enhancing the diagnostic potential of magnetic resonance imaging, offering promising improvements in accurately identifying brain tumors and differentiating different tumour types (
124,
125).
DIAGNOSTIC USES OF MOLECULAR TECHNIQUES (PCR, FISH, DNASEQUENCING) IN VETERINARY ONCOLOGY
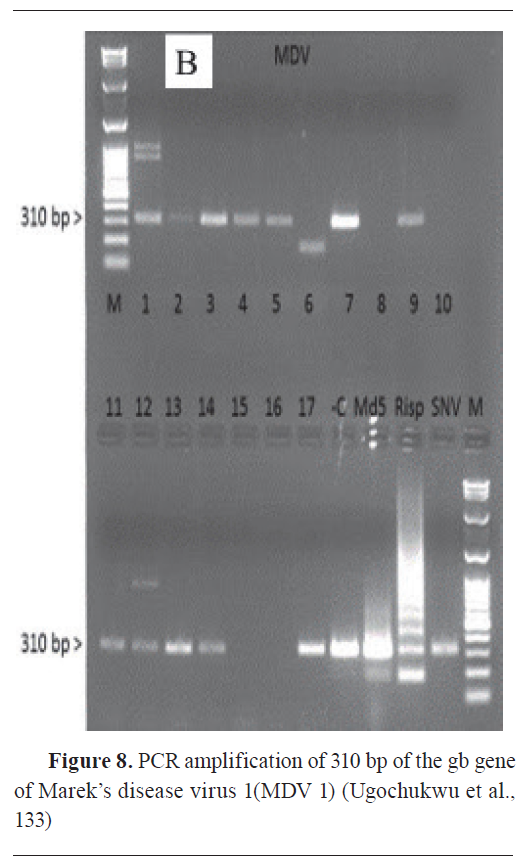
Molecular techniques, such as polymerase chain reaction (PCR) (
Fig. 8), Fluorescence in situ hybridization (FISH), and deoxyribonucleic acid (DNA) sequencing, are integral to advancing diagnostic capabilities in veterinary oncology. They offer precise methods for analyzing genetic and molecular alterations in animal tumours, providing comprehensive insights into the molecular landscape of cancers (
37,
51). PCR, a widely used method, amplifies specific DNA sequences, aiding in the detection of genetic mutations associated with veterinary tumours. It identifies specific genetic markers, contributing to accurate diagnosis and targeted treatment (
38,
126).
Fluorescence in situ hybridization (FISH) visualizes and detects DNA sequences within tumour cells, identifying chromosomal abnormalities and gene rearrangements. This technique enhances understanding of tumour biology and aids in cancer classification (
127,
128). DNA sequencing, including Sanger sequencing and next generation sequencing (NGS), allows comprehensive analysis of an animal’s genome, providing in-depth insights into genetic mutations in veterinary tumours. Next generation sequencing facilitates high-throughput sequencing, identifying multiple genetic changes simultaneously (
129,
130). These techniques contribute to early tumour detection by identifying specific genetic markers, enabling timely intervention. They aid in tumour classification, revealing unique molecular signatures for more accurate and personalized treatment strategies by optimizing treatment plans based on real-time molecular information (
129,
131). They also assist in identifying prognostic markers, offering valuable information about tumour aggressiveness and likelihood of recurrence, guiding treatment decisions and long-term care (
131).
Therefore, PCR, FISH, and DNA sequencing have revolutionized veterinary oncology by enabling early detection, accurate classification, and personalized treatment strategies. These molecular tools empower veterinarians to deliver precise and personalized care, ultimately enhancing the management and outcomes of cancer in animals.
Problems associated with use of molecular techniques (PCR, FISH, Sequencing) as diagnostic technique in veterinary oncology
Molecular techniques can be expensive and may require specialized equipment and expertise, limiting their availability in certain veterinary settings (
42,
129,
131). The success of molecular analysis in veterinary practice depends on the quality and integrity of the DNA/RNA extracted from the tissue. Degradation during sample handling or storage can lead to unreliable results (
132).
 DIAGNOSTIC USES OF SEROLOGICAL MARKERS IN VETERINARY ONCOLOGY
DIAGNOSTIC USES OF SEROLOGICAL MARKERS IN VETERINARY ONCOLOGY
Serological markers, also called tumour markers or biomarkers, are pivotal in veterinary oncology, contributing significantly to the diagnosis. They facilitate early tumour detection by revealing elevated levels before clinical signs manifest, prognosis, and monitoring responses to cancer treatments like surgery, chemotherapy, or radiationtherapy, with changing marker levels indicating treatment effectiveness or lack thereof (
5,
134). These markers, detectable in blood, serum, or other body fluids, exhibit altered levels in response to the presence of a tumour (
60,
135).
Serological markers provide additional information to help target specific areas for further investigation, which is particularly useful when imaging alone does not yield a definitive diagnosis. In research and clinical trials, serological markers are important as they contribute to the development of new diagnostic methods and treatment strategies in veterinary oncology (
131).
Problems associated with the use of serological markers as a diagnostic technique in veterinary oncology
The sensitivity and specificity of serological markers may vary, and false positives/negatives can occur, impacting the accuracy of tumour diagnosis (
136,
137). Cross-reactivity with other conditions or diseases may lead to misinterpretation of serological results. However, it’s important to note that while valuable, serological markers are not always tumour-specific, and factors like inflammation and other diseases can influence their levels. Therefore, interpreting serological marker results should be done in conjunction with other diagnostic tests and clinical observations (
138).
CONCLUSION
It is important for veterinary pathologists to consider these limitations and use a combination of these techniques to improve the accuracy of tumour diagnosis in animals. We suggested an infographic flowchart of a sequential diagnostic plan for veterinary oncologist (
Fig. 9). Additionally, advancements in technology (such as liquid biopsies, next generation sequencing (NGS), artificial intelligence and machine learning, nanotechnology, liquid biopsy-based multi-cancer early detection (MCED), optical coherence tomography and multiplexed immunoassays) and ongoing research may address some of these challenges over time. As these innovations in technologies could migrate from human medical practice into veterinary medical practice, they create an improved awareness and have clear understanding of their functionality and advantages will expedite their fast adoption, for the benefit of veterinary profession and their beloved patients.

In addition, the integration of these cutting-edge technologies in cancer diagnostics will enhance our ability to detect tumours earlier, characterize them more precisely, and monitor treatment responses effectively. These advancements will definitely pave the way for more personalized and effective neoplasm care, ultimately improving patient outcomes.
CONFLICT OF INTEREST
The authors declare that they have no financial or non-financial conflict of interest regarding authorship and publication of this article.
ACKNOWLEDGMENTS
The research was supported by the University of Nigeria NEEDS Assessment. Special acknowledgement is reserved for Late Professor Claudia Cafarchia of the Department of Veterinary Medicine, University of Bari for her words of advice and sharing from her wealth of experience in scientific writing during the writing of this manuscript.
AUTHORS’ CONTRIBUTION
ICIU, JNO, SJE, IL, MOE, and OAA were involved in the conceptualization, investigation, methodology, software, writing of the original draft, and review and editing of the paper. ICIU and IL performed formal analysis, supervision, validation, and final approval of the paper.

 10.2478/macvetrev-2026-0015
10.2478/macvetrev-2026-0015









