INTRODUCTION
Brucellosis is a serious bacterial zoonotic disease of global importance caused by bacteria of the genus
Brucella. It affects a variety of mammals, including humans, ruminants, swine, rodents, wild animals, and marine mammals. It was previously believed that there are just three major
Brucella species that are significant from a zoonotic standpoint,
Brucella melitensis (sheep and goats),
Brucella abortus (buffaloes and cattle), and
Brucella suis (pigs). Nowadays,
B. ceti (cetaceans),
B. pinnipedialis (pinnipeds) (
1) (both in marine mammals),
B. inopinata (humans-zoonotic importance) (
2), and
B. microti (voles) (
3) which is suspected to be infectious to man, are identified as other important
Brucella species.
B. papionis (baboons), and
B. vulpis (foxes) (
4) are still under question mark about their infectivity for humans. CDC (Centers for Disease Control and Prevention) classified
Brucella as a category-B pathogen that has the possibility to be one of the components of biological weapons, as it can be transmitted through aerosols (
5).
Brucella spp. is considered one of the most important and common causative agents for laboratory staff and workers (laboratory-acquired pathogens) (
6).
Brucellosis has no particular clinical characteristics, so diagnosis of brucellosis depends mainlyonlaboratory diagnosis, either microbiological culture for isolation and identification or serological tests such as the Complement Fixation Test (CFT), ELISA, and slide agglutination tests such as the Rose Bengal test, as well as molecular biology diagnosis. Slide agglutination tests, especially the Rose Bengal test are simple and rapid tests used as pilot tests for screening animal brucellosis. Anyhow, it is recommended that the RB test should be used with other confirmatory serological tests (with high sensitivity and specificity), such as CFT and I-ELISA, as a reference method in the absence of the main gold standard method (isolation and identification) (
7).
The aim of this study was to produce freeze-dried Rose Bengal (FD-RB) antigen and buffered acidified plate agglutination (FD-BAPA) antigen as a strategic store for emergency outbreaks, and to evaluate their stability, sensitivity, specificity, pH, PCV, and other performance criteria.
MATERIAL AND METHODS
Bovine serum samples Ninety-five bovine sera were randomly collected from non-vaccinated animals of infected flocks. Brucellosis-negative and -positive sera were determined by using the traditional Rose Bengal test as a pilot test for screening of
Brucella antibodies in ruminants (
8,
9,
10).
Rose Bengal antigens (RB antigen) and buffered acidified plate antigen (BAPA)
All slide agglutination antigens were prepared and performed according to OIE and Alton (
8,
10) in the Veterinary Serum and Vaccine Research Institute (VSVRI) using a reference strain,
Brucella abortus biovar 1 (S99).
Lyophilization using a freeze-drying method
A stabilizer (an additive preserving material) was prepared and used for the production of RB and BAPA antigens. These ingredients, which consist mainly of protein, polysaccharides, or both, were used in the following proportions: 1% sodium glutamate, 2.5% enzymatic digest of casein, and 5% sucrose (
11).
Physicochemical tests of all prepared antigens
Physicochemical tests were done in compliance with British Pharmacopoeia (2012) (
12). Physicochemical tests were done by periodical check of the appearance, negative pressure (vacuum), and solubility of the lyophilized cake with suitable diluents. Additionally, color, consistency, and presence of particles of the freeze-dried antigen product were checked (
13). In this study, in-house prepared freeze-dried antigens were reconstituted with a suitable volume of their acidic buffer and checked for PCV, pH, color, and absence of auto- agglutination.
Short-term stability study
The freeze-dried antigens were assessed in five periods (once/per month) after the production date to confirm their short-term stability. In each period, quality control and performance characteristics assessment was performed.
In-house smooth Brucella lipopolysaccharide (S-LPS, Hot Saline Extract)
According to Alton and Plackett (
10,
14), S-LPS was prepared and checkerboard titrated to be used as an Indirect Enzyme-Linked Immunosorbent Assay (I-ELISA) coating antigen (
10). Home-made I-ELISA was carried out using 96-well polystyrene flat-bottom micro plates according to the procedure described by OIE and Alton (
8,
10). All ELISA buffers used in this study were previously validated. The OD values were measured at 450 nm by using an ELISA Plate Reader (Thermo Scientific Multiskan FC, United States) and TMB as a substrate. This in- house ELISA was validated in comparison with the IDEXX Brucellosis Serum Ab Test for Bovine Brucellosis, measuring Linearity, Specificity (cross-reactivity), Sensitivity and Intra-assay precision using high, moderate, and low positive and negative
Brucella-infected bovine sera.
Validation of in–house ELISA
Validation of ELISA was done by measuring Linearity, Specificity (cross-reactivity) using serum against gram-negative bacteria that cross-reacted with serological tests for diagnosis of brucellosis, Sensitivity and Intra-assay precision.
Warm complement fixation test (CFT)
Hemolysin and complement were prepared, titrated, and preserved according to Alton (
10) compliant with Hennager (
15) and Stowell, H.E., (personal communication, November 22, 2010). The test was performed according to the American SOP described by (
16). The cutoff point of positive CFT is ≥20 international CFT units (ICFTU)/c.
All quality control and quality assurance for serological tests were fulfilled according to the requirements of OIE guidelines and ISO/IEC 17025:2017 (
8).
The evaluation of the test performance was carried out by calculating sensitivity, accuracy, positive predictive value or precision, negative predictive value, relative sensitivity, relative specificity, false positive rate, and false negative rate in addition to the probability ratio of positive results and the likelihood ratio of negative results. All the above-mentioned analyses were conducted by using (
http://vassarstats.net/clin1.html, https://ebm-tools.knowledgetranslation.net/calculator/diagnostic/ and
http://onlineconfusionmatrix.com/). Receiver Operating Characteristics (ROC) graph was plotted for cut-off selection at its best accuracy. ROC was generated to determine accuracy, specificity, and sensitivity of the tests by using CFT as a reference test. ROC was conducted using the statistical software MEDCALC. The logical relation of the reference test (CFT) with other serological tests is determined by a Venn diagram. The expressed AUROC represents accuracy. The higher the AUC, the more the accurate the test. ROC curve was constructed for each slide agglutination antigen, and optimal thresholds were calculated based on the optimized sensitivity and specificity.
Agreement between all serological tests performed in this study was calculated by generating a Venn diagram by using online platform https://
www.statskingdom.com/venn-diagram-maker.html.Ethical approval and consent for participation
The study was approved by ethical committee of CLEVB (arc.clevb.51429).
RESULTS
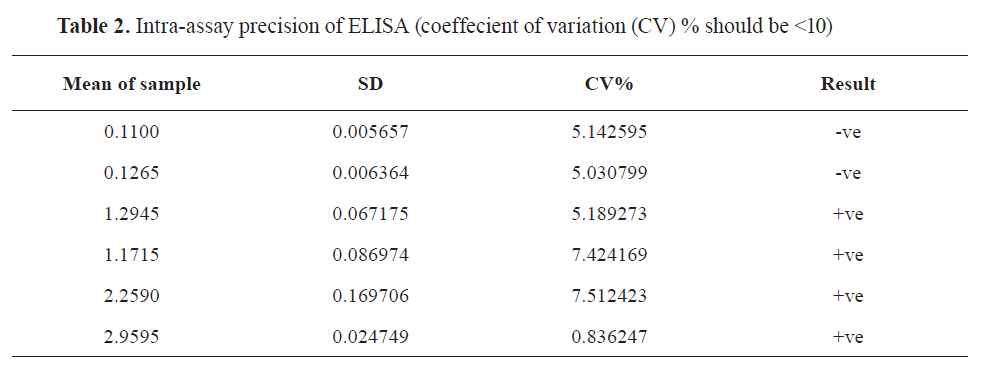
Validation of in–house ELISA
All parameters were valid and satisfactory (
Fig. 1 and
Tables 1 and
2).
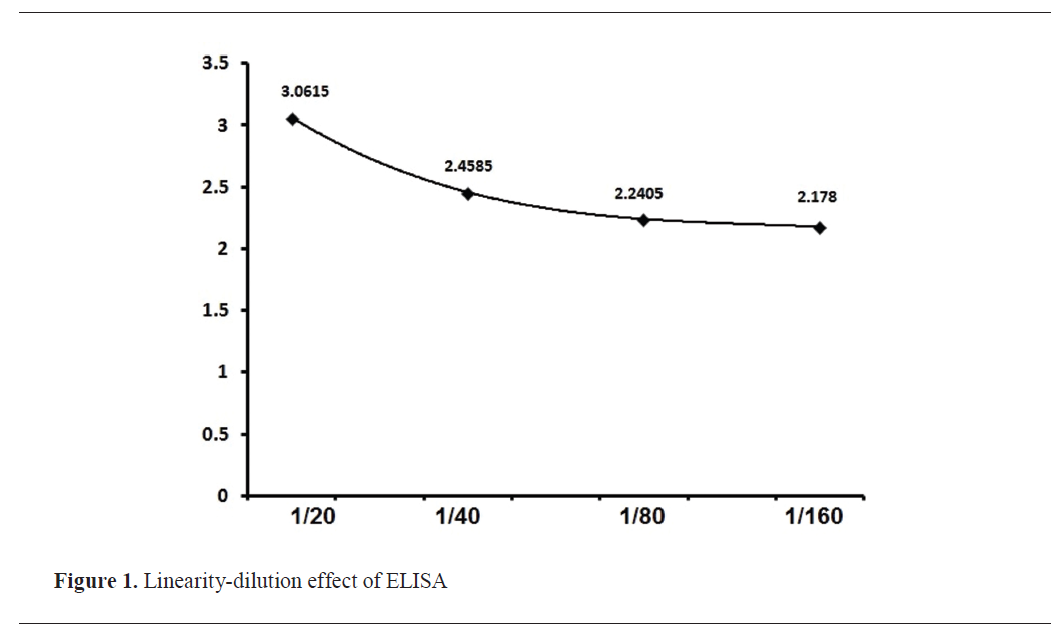

Rose Bengal antigen was with PCV 8%, pH 3.65, and rose color, while BAPA antigen was with PCV 11%, pH 3.7, and blue color. Diagnostic performance properties, including AUROC (Area Under the Curve), specificity, sensitivity, PPV, and NPV, were within 95% CI.
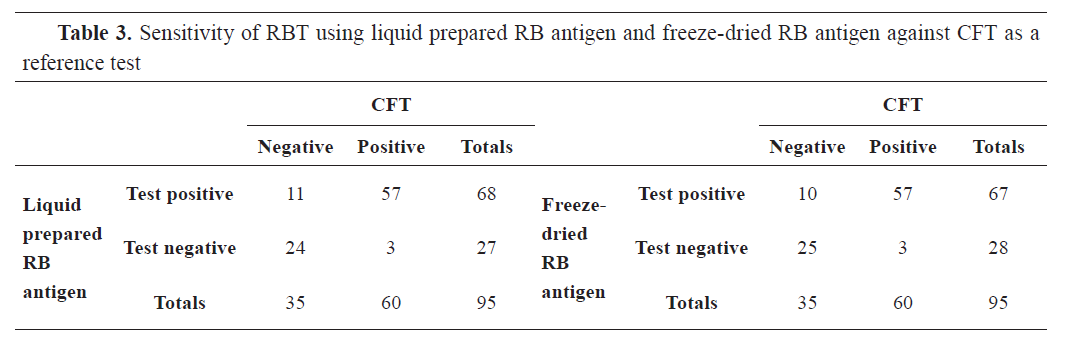
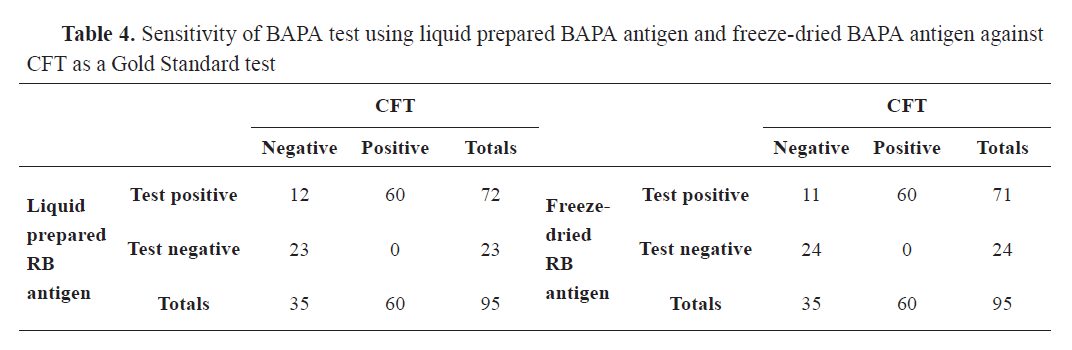
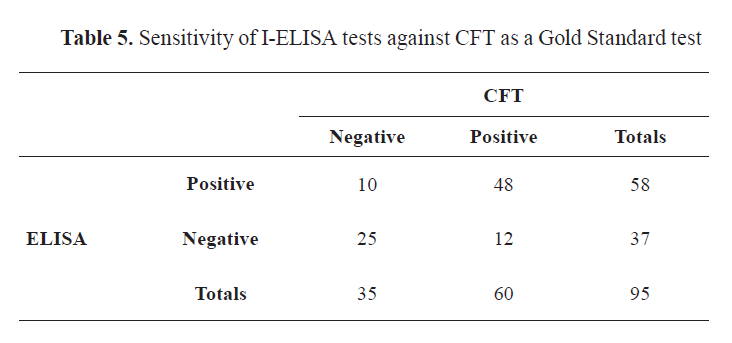
Out of 95 bovine sera tested for brucellosis, the Rose Bengal plate test was positive by Liquid Rose Bengal (LRB) and freeze-dried Rose Bengal (FDRB) in 68 (71.58%) and 67 (70.53%) samples, respectively. Buffered Acidified Plate Antigen (BAPA) was positive in 72 (75.79%) and 71 (74.74%) of the samples when tested by Liquid Buffered Acidified Plate Antigen (LBAPA) and Freeze-Dried Buffered Acidified Plate Antigen (FDBAPA), respectively. I-ELISA was positive for 58 (61.05%), while CFT for 48 (50.53%) samples (
Tables 3,
4, and
5).
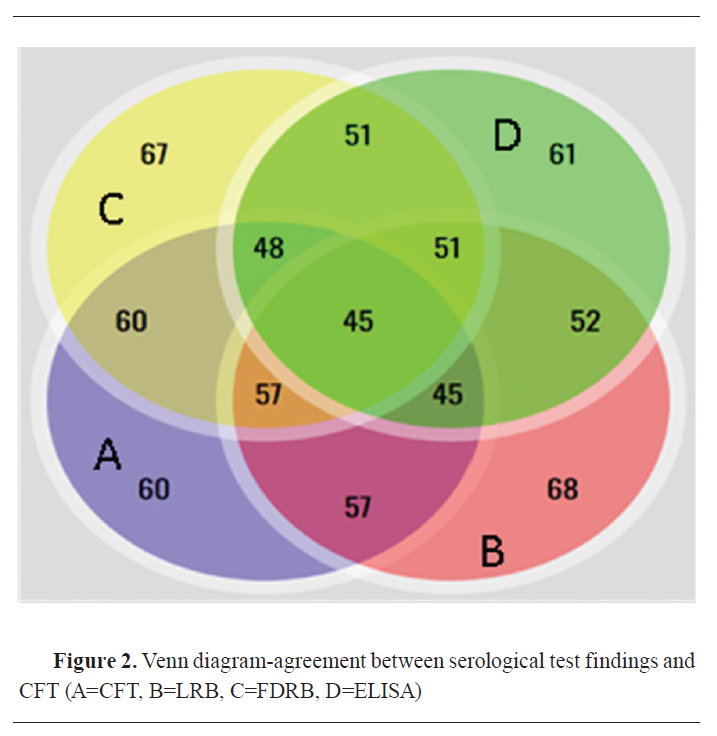
The reactors’ agreement with the reference test (CFT) was 57 (60%) for LRB and FDRB, 60 (63.16%) for LBAPA and FDBAPA, and 73 (76.84%) for I-ELISA (
Fig. 2 and
3).
A Venn diagram (
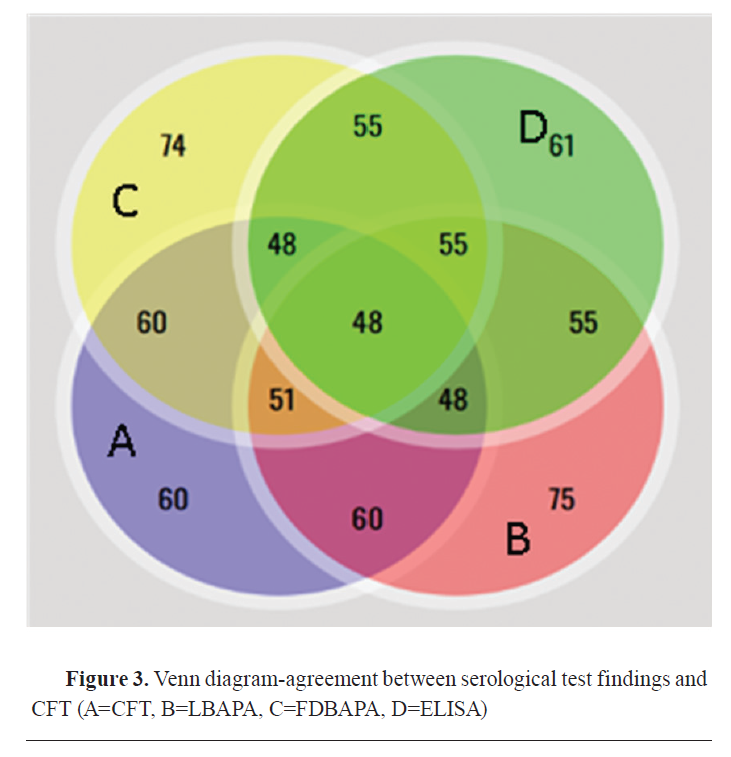
Fig. 2) expressed in detail the agreement of positive results among CFT, LRB, FDRB, and ELISA. There was no positive case detected by CFT. Both RB antigens detected 4 positive cases, single positive case was detected by LRB and ELISA, and 3 positive cases were detected by ELISA. While A Venn diagram (
Fig. 3) expressed in detail the agreement of positive results among CFT, LBAPA, FDBAPA, and ELISA.
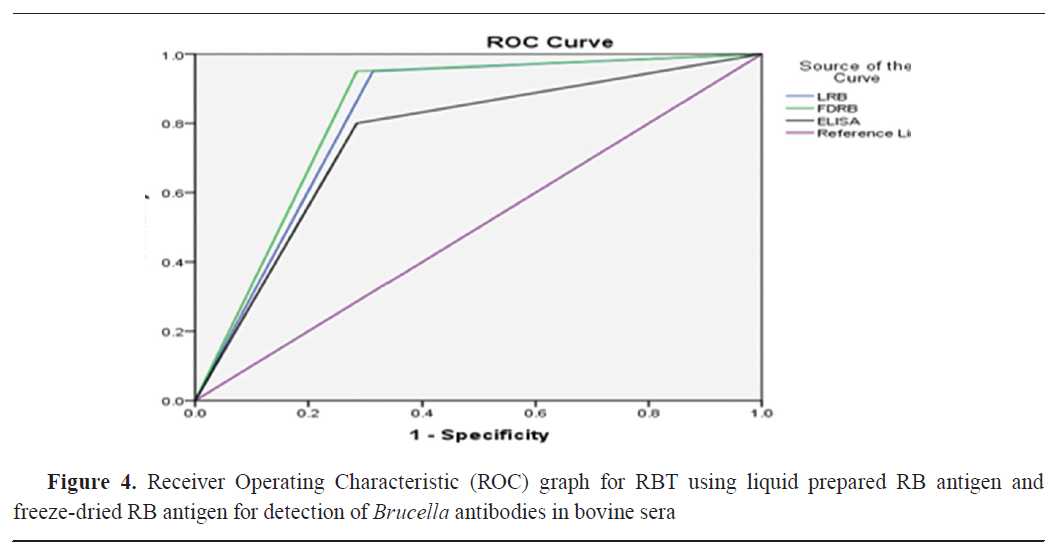
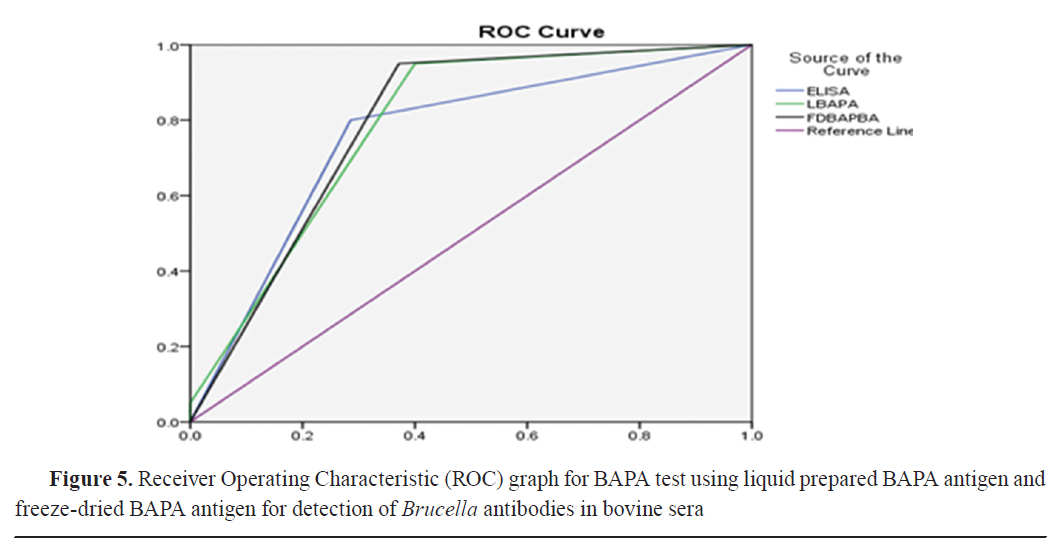
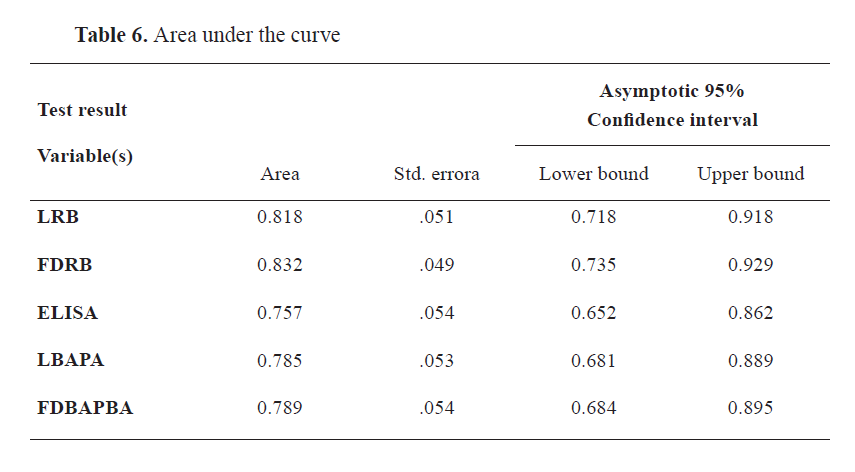
AUROC values were 0.818, 0.832, 0.757, 0.785, and 0.789 for LRB, FDRB, ELISA, LBAPA, and FDBAPBA, respectively (
Fig. 4 and
5 and
Table 6).
The ROC curve showed similar specificity and sensitivity as the in-house prepared antigens and the ELISA assay by using the cut-off points with consideration of CFT as the gold standard. The AUC analysis of all slide agglutination tests and I-ELISA ranged between 75.7 and 83.2%. The highest AUROC values were for FDRB, which indicated its good efficiency in the diagnosis of brucellosis, while the lowest AUROC was for I-ELISA. Тhe AUROC values for LRB, FDBAPA, and LBAPA were 81.8, 78.9, and 78.5%, respectively. Based on the ROC curve, the difference between the two rose Bengal and the two BAPA antigens was 0.014 and 0.004, respectively. These differences were non- significant. Rose Bengal had higher accuracy than BAPA and ELISA.
Diagnostic performance characteristics, including sensitivity, relative sensitivity, relative specificity, prevalence, true positive, false positive, true negative, false negative, NPV, FPR, FDR, FNR, accuracy, MCC, LR+, and LR- ,were calculated as shown in
Table 7.
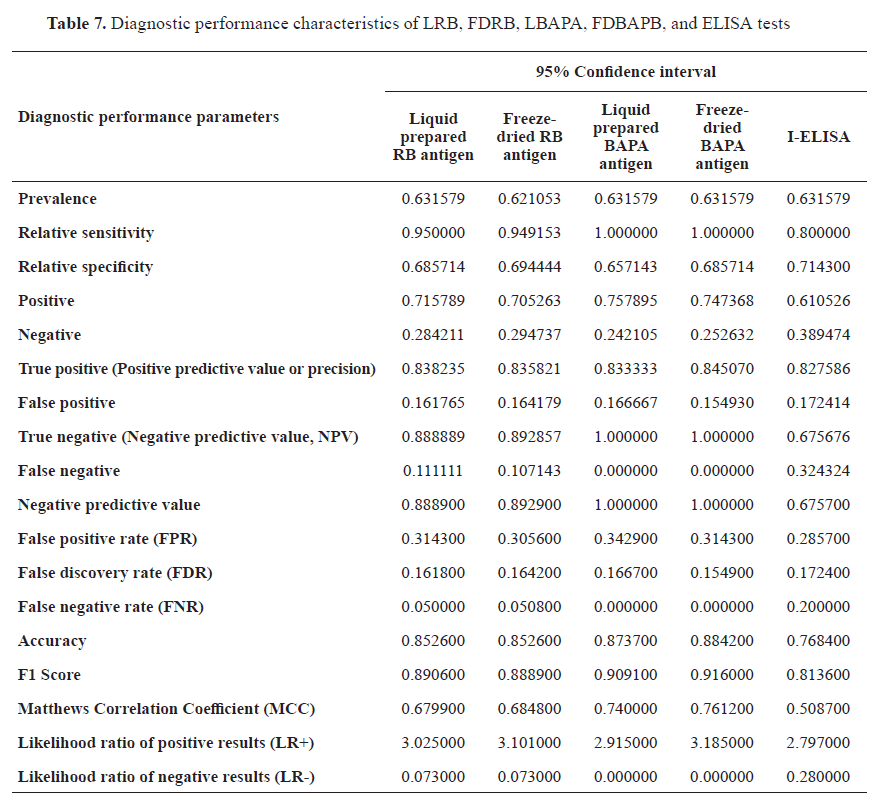
Anyhow, I-ELISA showed the highest percentage of false positive, false negatives and false discovery rate of false negative rates, which had a negative impact on the accuracy of diagnostic results, while the others had a positive impact and increased sensitivity and specificity of these tests with more accurate results. Results revealed that freeze-dried and liquid-prepared antigens were with approximately the same diagnostic performance characteristics and showed no significant difference.
Physicochemical tests and short-term stability
The obtained results indicated that there was no collapse (shrinkage) of lyophilized dry cake in the bottles. Both antigen batches were stable and fulfilled the recommended specifications described by Blasco (
18) with no evidence of increased moisture content. This stability was observed 5 months post-production. Lyophilized antigens showed good solubility in acidic (lactic acid) diluent under negative pressure (vacuum) observed throughout longer period. The intensity of the red and blue color stains of rose Bengal and BAPA antigens was stable and suitable for the test. Also, the PCV after reconstitution of the recommended volume of diluents was about 8% and 11% for RB and BAPA antigens, respectively. There was no autoagglutination observed between the control and field
Brucella-positive and
Brucella-negative sera.
DISCUSSION
Brucellosis is one of the most important worldwide zoonotic diseases affecting the public health and economy. The prevalence of the disease is related to numerous factors. The capability of a country to finance prevention, the implementation of control programs, and the hygienic management practices of the farm are considered among the most significant factors affecting the prevalence. As clinical signs are not characteristic for the disease, it is mainly diagnosed by utilizing laboratory methods which are based on isolation of the etiological agents on specific media and identification by biochemical, morphological, serological, and culture methods. The most common molecular diagnostic method is the BRUCELADDER assay, which is based on genetic identification. However, the gold standard for laboratory diagnosis of brucellosis is isolation and identification of the bacteria. The serological detection of specific antibodies in bovine and ovine samples is commonly performed by Rose Bengal Plate Test (RBPT) and Complement Fixation Test (CFT) which are commonly accepted in the global animal trade (
8). CFT is used as a gold standard test in the absence of isolation and identification of the causative agent (
17). The joint FAO/WHO expert committee on brucellosis has supported the use of the RBPT as a pilot screening test for the diagnosis of
B. melitensis infection in sheep and goats (
9,
18,
19). RBPT is a commonly used approach for diagnosis and for determining the prevalence since it is affordable, reliable, and quick (
20).
More than 30 years ago, the RB test was created and used for diagnosis of bovine brucellosis. The standardization of the antigen and the used strain is a significant factor that affects the sensitivity of the slide agglutination test. According to the EU regulations (Council Directive 64/432/EEC, 1964), The International Standard anti-
B. abortus serum (ISaBS) must be diluted 1:47.5 (21 IU/mL) in acidic lactate buffer at pH 3.65 ± 0.05 to achieve positive agglutination. Dilution rates of 1:55 (18.2 IU/mL) are used to produce negative reaction.
The antigen standardization for the American Rose Bengal antigen and BAPA relies on the packed cell volume (PCV) which must be 8% and 11%, respectively (
10,
21). These standardization conditions appear to be appropriate for the diagnosis of
B. abortus infection in cattle. However, they also limit the sensitivity of the test, which results in decreased performance for the diagnosis of
B. melitensis infection in sheep (
9,
18).
This clarifies that some commercial RB antigens have a relatively low sensitivity for diagnosing brucellosis in sheep and goats (
9,
18). It also explains why a large percentage of sheep and goats from
B. melitensis-infected areas have negative RB test results but positive CF test results (
9).
These events have prompted serious concerns about the effectiveness of the RB as a standalone test in small ruminants. The RBPT is an internationally recognized test for screening of brucellosis by identifying particular IgG antibodies, and to a lesser extent IgM (the acidity of the Rose Bengal antigen breaks down the disulfide bond in IgM) (
22,
23). The test is straightforward and easy to conduct, and it doesn't require any specialized laboratory facilities. Positive results need to be confirmed by CFT or ELISA because it is a qualitative test only (
24,
25).
This study established very good agreement between lyophilized and liquid-prepared slide agglutination antigens. The agreement is determined by diagnostic performance parameters and the area under the curve (AUC) in ROC.
ELISA showed higher specificity than RB and BAPA tests. These findings are in partial agreement with the reports by Legesse and Šerić-Haračić (
26,
27). RB and BAPA showed higher sensitivity than ELISA in the current study compared to these reports. This difference could be explained as a result of a different approach for preparing the agglutination antigens. The referenced reports were preparing them for routine monitoring, and the antigens for the current study were prepared for validation purposes from bovine sera which were tested before being utilized for the production of the Rose Bengal antigen. However, it should be noted that high sensitivity may be attributed to false- positive findings as a result of cross-reactivity between antibodies against
Yersinia enterocolitica type 0.9 and
Escherichia coli 0:157H, or other gram-negative bacteria that possess O-side chain lipopolysaccharide identical to that of smooth
Brucella species.
Anyhow, three different diluents were used in this research article during freeze-drying; the first were Rose Bengal and BAPA diluent buffers (
10), the second was PBS, and the third was stabilizer according to Angus (
11). Poor stability was observed with diluent buffer and PBS manifested as lyophilized cake and strong auto-agglutination one month post lyophilization. High stability was observed when using stabilizer which achieved reconstitution of the diluent buffer.
The Stability Study is performed to assess the capacity of biological products to retain its physical, chemical, biological, and microbiological properties throughout its shelf life (
28).
Lyophilized antigens are a combination of chemical (non-biological) and biological ingredients that are sensitive to numerous environmental factors and changes in the chemical components. The stability of lyophilized products could be influenced by numerous factors, such as the composition of the stabilizer, reconstitution diluents, heavy water, the process and equipment used in the production, lyophilization programs, the cold chain used for storage and maintenance, and the products’ transportation (
29). In this study, short-term stability was carried out along 5 months after the production of the lyophilized antigens by utilizing Roux bottle methods as described in OIE and Alton (
8,
10). Other factors are the reconstitution process and diluents as well as storage conditions, which are important to establish the shelf life (expiry) (
30). All stability parameters were valid, which suggests long-term stability. Commercial Rose Bengal antigen had an expiry date of about one year, while this study suggests that freeze- dried antigens can stay with long stability for years, if stored in lyophilized state without reconstitution. This finding should be confirmed with long-term stability studies.
CONCLUSION
It is concluded that the newly prepared freeze- dried slide agglutination antigens were approximately as efficient as traditionally prepared antigens by considering the sensitivity and specificity for detection of brucellosis. The high short-term stability suggests that they may be stored safely and could be used reliably during emergency or other types of disease outbreaks. Anyhow, further investigation and examination in the context of long-term stability have to be done to determine the duration of stability and expiry date (shelf life), which is one of the goals of this method of production: to be capable of storing this antigen for a long period while keeping its diagnostic potential.
CONFLICT OF INTEREST
The authors declare that they have no financial or non-financial conflict of interest regarding authorship and publication of this article.
ACKNOWLEDGMENTS
The authors acknowledge the Central Laboratory for Evaluation of Veterinary Biologics (CLEVB) and Veterinary Serum and Vaccine Research Institute (VSVRI) for their cooperation.
AUTHORS’ CONTRIBUTION
LFF formulated and designed the study. MAE, MSD, and LFF were included in experimentation and laboratory work. WSS and AMM collected reviews, followed up the work and interpretations. WSS supervised and wrote the original manuscript. MAE edited and revised the manuscript. All authors have read and approved the final manuscript.

 10.2478/macvetrev-2026-0016
10.2478/macvetrev-2026-0016











